- Startpagina tijdschrift
- Volume 15 (2011)
- numéro spécial 1
- Speciation of arsenic and mercury in feed: why and how?
Weergave(s): 9263 (26 ULiège)
Download(s): 279 (1 ULiège)
Speciation of arsenic and mercury in feed: why and how?

Abstract
The understanding of the mechanisms of biological activities and biotransformation of trace elements such as arsenic and mercury has improved during recent years with the help of chemical speciation studies. However, the most important practical application of elemental speciation is in the area of toxicology. Toxicological knowledge on the individual trace element species can lead to more specific legislation of hazardous substances found in feed. Examples here are arsenic, where the inorganic forms are the most toxic, and mercury, where the organic form methylmercury is more toxic than inorganic mercury. In the present paper an overview of the current knowledge on arsenic and mercury speciation in feed and analytical methodologies for arsenic and mercury speciation analysis are given. Additionally the current status and expected future developments within legislation for trace element speciation in feed as well as initiatives for the establishment of standardized methods for determination of inorganic arsenic and methylmercury are presented.
Inhoudstafel
1. Introduction
1Formulated feed is the main source of most chemical contaminants in farmed fish, where the type of contaminant depend on the type of feed. In fish meal contaminants such as heavy metals and metalloids (mercury and arsenic) may be present, whereas in fish oil unwanted substances such as dioxins, dioxin-like PCB’s and brominated flame retardants may be present. If the feed is based on ingredients of vegetable origin unwanted genetically modified ingredients (GMOs) may have been used, and metals like lead and cadmium can be present in the non-lipid phase, whereas in vegetable oils pesticides may occur. In the end all of the contaminants listed end up affecting the quality and safety of the feed and therefore the produced animal.
2The maximum allowed concentrations for the total content of the heavy metals arsenic, lead, cadmium and mercury in feed are regulated in European Directive 2002/32/EC (European Communities, 2002) and amendments. Consequently, traditional feed control measures have only included measurement of total amounts of these metals. However, the biological activity, mobility, bioavailability and also the toxicity of an element also depend on the chemical form in which the element is present. Thus in order to achieve correct information concerning these factors, analytical procedures that are able to distinguish between the different chemical forms are required, i.e. speciation analysis. A definition of chemical species, speciation and speciation analysis was suggested by Templeton et al. (2000).
3Speciation definition: A chemical species is a specific form of an element defined as to isotopic composition, electronic or oxidative state, and/or complex or molecular structure. Speciation of an element is the distribution of an element amongst defined chemical species in a system. Finally speciation analysis is defined as the activities of identifying and/or measuring the quantities of one or more individual chemical species in a sample.
4The most important practical application of speciation is undoubtedly within the field of toxicity (Proust el al., 2005). Although legislators have become more aware of the importance of speciation, widespread implementation of maximum levels on chemical species has still not taken place, partly due to lack of detailed toxicological data on species level as well as available validated and reliable analytical methods suitable for routine control analysis.
5The aim of the present paper is to provide an introduction to heavy metal speciation within the area of feed analysis together with a short overview of the status regarding analytical speciation techniques. The focus will be on the elements arsenic and mercury as these elements presently attract most attention with respect to feed safety and feed control measures.
2. Speciation requirements
2.1. Speciation of arsenic
6Arsenic has a very complex chemistry illustrated by the more than fifty different naturally occurring arsenic containing chemical species that have been identified, mainly in samples from the marine environment (Francesconi et al., 2004). Table 1 shows the names and chemical structures of a range of arsenic species present in biological samples. Inorganic arsenic comprises the two oxyanions arsenite As(III) and arsenate As(V). They are readily interconverted and therefore often found together, with As(V) being thermodynamically favored under normal environmental oxygen levels (Cullen et al., 1989). Both species are usually reported in marine animals as minor constituents, usually less than 5% of the total arsenic (Edmonds et al., 1993). However, some exceptions have been reported including certain types of algae (e.g. Hizikia fusiforme approximately 70% inorganic arsenic) (Almela et al., 2002) and certain bivalve samples from Norway (up to 42% inorganic As) (Sloth et al., 2008).
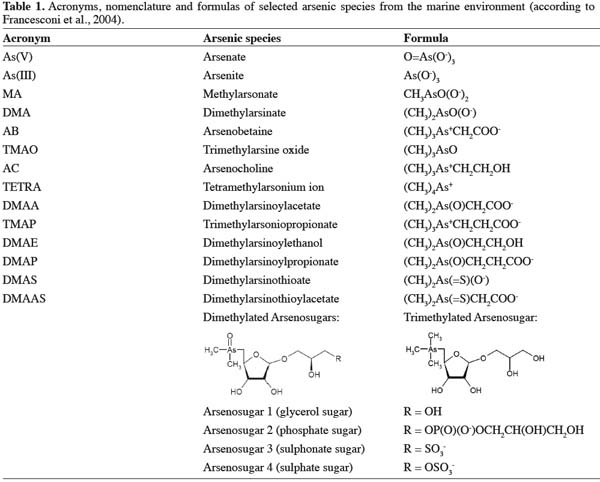
7Simple methylated arsenic species with 1-4 methyl groups incorporated in the molecule such as monomethylarsonic acid (MA), dimethylarsinic acid (DMA), trimethylarsine oxide (TMAO) and tetramethylarsonium ion (TETRA) are metabolites from the same biogenetic pathway involving reduction and methylation of As(V) and are often found together (Challenger, 1945). A whole range of different organoarsenic compounds exists. Arsenobetaine is the predominant species in fish and crustaceans usually constituting more than 80% of the total arsenic content (Francesconi et al., 1997), whereas other species like arsenocholine (AC) and trimethylarsoniopropionate (TMAP) are usually only minor constituents in marine animals. An interesting group of arsenic species (the so-called arsenosugars) dimethylated and trimethylated arsenic-riboside compounds have also been found at low concentrations in various marine animals, but their origin have in most cases been suggested to be related to algae present in the food chain (Francesconi et al., 1997), and these compounds are typically found at high concentrations in marine algae. A novel group of arsenic compounds were recently discovered, namely the thioarsenicals, in which arsenic is bound to sulphur and until now approximately 9 compounds have been identified (Kahn et al., 2005; Raml et al., 2005). Arsenic may also be present in lipid-soluble compounds, so-called arsenolipids (Taleshi et al., 2010). Analysis of fish oils for feed production showed total arsenic concentrations in the range of 9.1-13.5 mg.kg-1 and it was estimated that approximately one third of the arsenic present in marine-based complete feedingstuff can be lipid bound (Sloth et al., 2005a). Since seafood is the main dietary source of arsenic for humans (EFSA, 2009) speciation of this element is crucial in order to provide relevant input on both feed and food safety questions.
2.2. Speciation of mercury
8Mercury is generally considered to be among the highest priority environmental pollutants on the global scale and it causes continuously concern as a contaminant in both feed and food. Mercury is one of the most highly bioaccumulated elements in the food chain (especially aquatic) and has been targeted for possible emission control by international agencies and organizations. Mercury exists in the environment as elemental mercury (Hg0), inorganic mercury and organic mercury (primarily methylmercury) (Horvat et al., 2005). Elemental mercury is referred to as mercury vapor when present in the atmosphere or as metallic mercury when present in liquid form. It has a high vapor pressure and is slightly soluble in water. It has a high lipophilicity, which makes it dissolve easily in lipid compartments. A wide range of inorganic mercury (Hg2+) salts exist, of which most are readily soluble in water. An exception is cinnabar (HgS) with a very low solubility (0.00001 g.l-1) (Simon et al., 2002). The high affinity of Hg2+ to S-groups provides an explanation of the toxicity mode of action of Hg2+. Interestingly the affinity for SeH groups is even higher, explaining the proposed antagonistic effect of selenium on mercury intoxication (Yoneda et al., 1997). Organic mercury compounds comprise in practice alkyl-, aryl- and alkoxymercury compounds, where the two latter groups are labile and biodegrade relatively easy, whereas alkyl compounds are more resistant. Monomethylmercury (MeHg) can bioaccumulate and biomagnify in aquatic food chains, resulting in exposure to the seafood eating part of the population. Indeed seafood is the main dietary source of mercury in the European population (SCOOP, 2004).
3. Feed safety
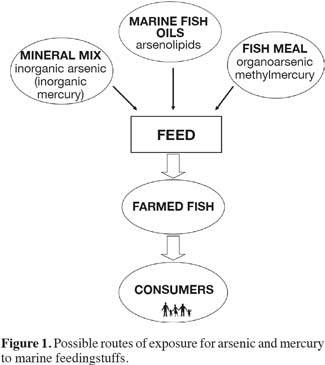
9Figure 1 illustrates the routes of arsenic and mercury compounds from marine feed ingredients to the final complete feed. In marine oils the concentration of arsenic may be high due to the presence of arsenolipids (Sloth et al., 2005a). In fish meal organoarsenic compounds (e.g. arsenobetaine) generally predominate, although the presence of inorganic arsenic naturally in the meal cannot be ruled out. Fish meal is also the dominant source for methylmercury in the fish feed. Mineral mixes or other feed additives may also be a potential route for exposure to inorganic salts of arsenic and mercury present as contaminants in minerals used to produce this type of ingredient. The risk assessment of arsenic and mercury and their species will be briefly discussed in the following section.
3.1. Risk assessment of arsenic
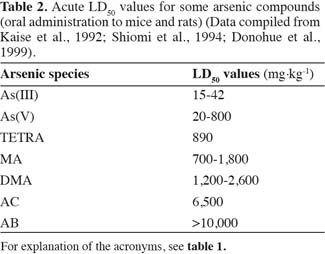
10To the general public, arsenic has the reputation as a potent poison stemming from the tasteless and odourless nature of arsenic trioxide, which for centuries has been viewed as an archetypical poison, being the choice for many homicidal and suicidal deaths. However, the toxicity of arsenic is highly dependent on its chemical form. Since the early work in the beginning of the last century, it had been accepted that most of the arsenic present in marine organisms was organic and non-toxic because animal studies showed that so-called fish-arsenic was non-toxic to rats and rapidly excreted in the urine (Coulson et al., 1935). The LD50 values of various arsenic compounds in table 2 show that the inorganic forms of arsenic are the most acute toxic, whereas MA and DMA show intermediate acute toxicity, and the trimethyl and tetraalkyl-arsonium compounds may be considered innocuous. In general, trivalent arsenicals are more toxic than their pentavalent counterparts. To date no acute toxicity has been reported for arsenosugars (Andrewes et al., 2004) and so far no studies on the toxicity of arsenolipids have been reported.
11One thing is the acute effects from single high doses of arsenic compounds; another is the long-term exposure and here inorganic arsenic has attracted a lot of focus. Chronic exposure to arsenic (e.g. via drinking water from areas of the world with very high inorganic arsenic levels in the well water such as in the Bangladesh region) may result in skin lesions, hypo- and hyperpigmentation (Blackfoot Disease) and vascular diseases with gangrenous changes. Other symptoms associated with chronic arsenic exposure are peripheral neurophathy, encephalopathy, altered heme metabolism, hepatomegaly, bone morrow depression, diabetes and renal function impairment (papillary and cortical necrosis) (NRC, 1999; Ng et al., 2003). Inorganic arsenic is a carcinogenic substance and United States Environmental Protection Agency has estimated a cancer slope factor for inorganic arsenic of 1.5 (mg.kg-1 per day)-1, which can be used to estimate the cancer risk associated with exposure to inorganic arsenic (US EPA, 1998). The Joint Food and Agriculture Organization (FAO)/World Health Organization (WHO) Expert Committee on Food Additives (JECFA) established in 1988 a Provisional Tolerable Weekly Intake (PTWI) value of 15 µg.[kg body weight (bw)]-1 per week for inorganic arsenic – corresponding to an intake of approximately 150 µg per day for an adult of 70 kg throughout the lifetime without adverse effects (WHO, 1988). However, this PTWI value was recently overruled by the European Food Safety Authority (EFSA), who published an opinion on arsenic in food and proposed a range of benchmark dose lower confidence limit (BMDL01) values between 0.3 and 8 µg.(kg bw)-1 per day for cancers of the lung, skin and bladder, as well as skin lesions. The estimated dietary exposures to inorganic arsenic for average and high level consumers in Europe are within the range of the BMDL01 values identified, and therefore there is little or no margin of exposure leading to a possible risk to some consumers (EFSA, 2009).
12Arsenic has been found in much higher concentrations in marine animals and algae compared to freshwater fish and terrestrial biota, due to marine organisms are capable of bioaccumulating arsenic by a factor of 100.000 compared to seawater organisms (Sloth et al., 2005b). As previously mentioned the non-toxic species arsenobetaine is in most cases the predominant arsenic species in marine animals and the content of the most toxic forms namely inorganic arsenic in various fish and seafood is usually below 5% (Uneyama et al., 2007). This was also demonstrated to be the case for marine feed by Sloth et al. (2005a), where 13 complete fish feed samples all had inorganic arsenic levels below 1.2% of the total arsenic contents.
13The maximum levels for the total arsenic content in feed have recently been amended in European Directive 2009/141/EC (European Commission, 2009) (Table 3). However, complete feedingstuffs for fish and also fish meal samples with high levels of total arsenic are at risk of being withdrawn from the market due to the EU maximum level for total arsenic even if the content of the toxicologically most relevant species namely inorganic arsenic is low. In a footnote in the EU directive is it stated the competent authorities can request an analysis to demonstrate that the inorganic arsenic content is less than 2 ppm for some of the products, including palm kernel expeller, marine feedingstuffs, seaweed and complete feedingstuffs for fish and fur animals. It is highly recommendable that the existing maximum level for total arsenic should be re-evaluated and future maximum levels should be based on the species of toxicological relevance, inorganic arsenic rather than total arsenic. Likewise, it is also recommendable that any future international legislation on arsenic in foodstuffs should be based on inorganic arsenic rather than total arsenic.

3.2. Risk assessment of mercury
14The toxicity and toxicokinetics of mercury in animals and humans depend on its chemical form. The kidneys are a target tissue for retention of mercury in populations exposed to inorganic mercury compounds or mercury vapor. Organic mercury compounds and mercury vapor may pass the blood-brain and placental barriers and here the brain is a target organ (Horvat, 2001). Among the organic forms, the most toxic is methylmercury (Horvat, 2001). Methylmercury is considered to be the most toxic due to the irreversibility of its effects on the central nervous system, which is the primary site of mercury toxicity in animals and humans. In humans, effects on neurological development have been observed in children of mothers, who orally have been exposed to methylmercury. Since methylmercury bioaccumulate and biomagnify in the aquatic food chain; long-lived carnivorous fish and marine mammals have the highest contents. As a consequence food authorities throughout the world have issued restrictive advice on the consumption of predator fish, especially for children and pregnant women. WHO has established a PTWI value for mercury of 5 µg.(kg bw)-1 per week and 1.6 µg.(kg bw)-1 per week for methylmercury (WHO, 2003). The difference in these values by a factor of three demonstrates the need for speciation analysis to be able to perform a correct risk assessment.
15The concentration of total mercury in feed is regulated in the legislation (European Commission, 2010) with maximum level of 0.1 mg.kg-1 for feed materials, 0.5 mg.kg-1 for feedingstuff produced by the processing of fish or other marine animals and 0.2 mg.kg-1 for compound feedingstuff for fish (Table 4). The most common source of mercury in feed materials is fish meal, where the predominant part of mercury will be present as methylmercury bound to the protein fraction. Data from the Norwegian feed monitoring programme gave mean total mercury levels around 0.05 mg.kg-1 (N > 300 samples) and a concentration range of 0.01-0.38 mg.kg-1 – all below the present maximum level (Maage et al., 2009). However, since methylmercury is recognized more toxic than inorganic mercury the determination of total mercury in feed may not always accurately reflect the risk posed by the organic mercury forms present. This was recently emphasized by EFSA, who in a recent opinion on mercury in feed called upon specific data on methylmercury in feed samples (EFSA, 2008).

4. Methods for speciation analysis
16Various analytical methodologies have been applied for speciation analysis of heavy metals in both feed and food. The analytical methods can be divided into a separation part and an element-selective detection part. Mainly liquid chromatography (LC), gas chromatography (GC) and capillary electrophoresis have been used for the separation of the species prior to detection. Various kinds of separation approaches for LC have been applied: size exclusion, ion exchange, reversed phase and ion pairing. The detection of the element of interest is subsequently performed by element-selective detectors [e.g. ICP-MS (Inductively Coupled Plasma Mass Spectrometry) or AF (Atomic Fluorescence)]. Most commonly used is the coupling of HPLC to ICP-MS. Although this approach is used in many laboratories, there is still a need for development of new, fast and inexpensive methods for speciation of both arsenic and mercury to be used in routine laboratories for future monitoring and control in both food and feed. These laboratories may not have expensive instrumentation like HPLC-ICP-MS available. The methods need to be simple, robust and easy to use in order to be applicable in routine control laboratories and preferably standardized. Several ongoing EU projects focus on the development of fast inexpensive detection methods. A European standard method for the determination of inorganic arsenic in marine-based feedingstuffs is under development by the European Committee for standardization (CEN), Technical Committee 327 “Animal feeding stuffs”, WG 4 concerning heavy metals, trace elements and minerals. The ongoing EU FP7 project entitled CONffIDENCE aims to further improve food safety in Europe by the development of fast and cost-efficient methods for the detection of a wide range of chemical contaminants in different food and feed commodities. CONffIDENCE focus on the heavy metals arsenic and mercury and the primary tasks include the development of off-line approaches for separation of inorganic arsenic from the organic arsenic species and separation of inorganic mercury from methylmercury, respectively, using SPE columns followed by element specific detection by Atomic Absorbance Spectrometry (AAS).
17The methods developed in the mentioned projects are expected to be collaboratively tested and will be ready to use for routine control purposes in the near future.
5. Conclusion
18The total concentration of trace elements does not always provide adequate information on bioavailability and toxicity and speciation analysis is required to obtain a correct risk assessment regarding trace elements in feed. There is a need for methods for future monitoring and control of heavy metals in both food and feed, which also takes the speciation of the metal into account. These methods need to be simple, inexpensive and robust and easy to use in order to be efficient for routine control laboratories and preferably standardized. HPLC- and GC-ICPMS are versatile tools for trace element speciation analysis, but also alternative options with off-line separations by SPE followed by detection by HG-AAS are inexpensive and simple speciation alternatives. Recently, EFSA released a new opinion on arsenic in food, which concluded that the PTWI from JECFA (WHO, 1988) is no longer appropriate and, in its assessment, which focused on more recent data showing effects at lower doses of inorganic arsenic leaving little or no margin of exposure for high/middle consumers. Hence, EFSA concluded that the possibility of a risk to some consumers could not be excluded (EFSA, 2009). The new assessment from EFSA may affect the legislation regarding arsenic in feed or food and emphasizes the need for simple methods for future monitoring.
Bibliographie
Almela C. et al., 2002. Heavy metal, total arsenic, and inorganic arsenic contents of algae food products. J. Agric. Food Chem., 50, 918-923.
Andrewes P. et al., 2004. Do arsenosugars pose a risk to human health? The comparative toxicities of a trivalent and pentavalent arsenosugar. Environ. Sci. Technol., 38, 4140-4148.
Challenger F., 1945. Biological methylation. Chem. Rev., 36(3), 315-361.
Coulson E.J., Remington R.E. & Lynch K.M., 1935. Metabolism in the rat of the naturally occurring arsenic of shrimp as compared with arsenic trioxide. J. Nutr., 10(3), 255-270.
Cullen W.R. & Reimer K.J., 1989. Arsenic speciation in the environment. Chem. Rev., 89(4), 713-764.
Donohue J. & Abernathy C., 1999. Exposure to inorganic arsenic from fish and shellfish. In: Chappell W., Abernathy C. & Calderon R., eds. Arsenic exposure and health effects. Oxford, UK: Elsevier Science Ltd.
Edmonds J.S. & Francesconi K.A., 1993. Arsenic in seafoods – human health aspects and regulations. Mar. Pollut. Bull., 26(12), 665-674.
EFSA, 2008. Mercury as undesirable substance in animal feed, scientific opinion of the panel on contaminants in the food chain. EFSA J., 654, 1-76.
EFSA, 2009. Scientific opinion on arsenic in food. EFSA J., 7(10), 1351-1550.
European Commission, 2009. Commission directive 2009/141/EC of 23 November 2009 amending Annex 1 to Directive 2002/32/EC of the European Parliament and of the Council as regards maximum levels for arsenic, theobromine, Datura sp., Ricinus communis L., Croton tiglium L. and Abrus precatorius L. Off. J. Eur. Union, L308, 20.
European Commission, 2010. Directive 2010/6/EU of 9 February 2010 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as regards mercury, free gossypol, nitrites and Mowrah, Bassia, Madhuca. Off. J. Eur. Union, L37, 29.
European Communities, 2002. Directive 2002/32/EC of the European Parliament and of the Council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Communities, L140, 10.
Francesconi K.A. & Edmonds J.S., 1997. Arsenic and marine organisms. Adv. Inorg. Chem., 44, 147-189.
Francesconi K.A. & Kuehnelt D., 2004. Determination of arsenic species: a critical review of methods and applications, 2000-2003. Analyst, 129, 373-395.
Horvat M., 2001. Mercury – Do we know enough? In: Ebdon L. et al., eds. Trace element speciation for environment, food and health. Cambridge, UK: RSC.
Horvat M. & Gibičar D., 2005. Speciation of mercury: environment, food, clinical and occupational health. In: Cornelis R. Handbook of elemental speciation II. Chichester, UK: Wiley.
Kahn M. et al., 2005. Two novel thio-arsenosugars in scallops identified with HPLC-ICPMS and HPLC-ESMS. Environ. Chem., 2, 171-176.
Kaise T. & Fukui S., 1992. The chemical form and acute toxicity of arsenic compounds in marine organisms. Appl. Organomet. Chem., 6(2), 155-160.
Maage A. & Julshamn K., 2009. Mercury, fish production, and aquaculture: past, present, and future. In: Bank M.S., ed. Mercury in the environment: pattern and process. Berkeley, CA, USA: UCA Press.
Ng J.C., Wang J. & Shraim A., 2003. A global health problem caused by arsenic from natural sources. Chemosphere, 52(9), 1353-1359.
NRC (National Research Council), 1999. Arsenic in drinking water. Washington, D.C., USA: National Academy Press, www.nap.edu/openbook/0309063337/html/R1.html.
Proust N., Buscher W. & Sperling M., 2005. Speciation and the emerging legislation. In: Cornelis R., ed. Handbook of elemental speciation II. Chichester, UK: Wiley.
Raml R. et al., 2005. Novel thioarsenic metabolites in human urine after ingestion of an arsenosugar, 2',3'-dihydroxypropyl 5-deoxy-5-dimethylarsinoyl-beta-D-riboside. Chem. Res. Toxicol., 18(6), 1444-1450.
SCOOP, 2004. SCOOP report 3.2.11. Assessment of the dietary exposure to arsenic, cadmium, lead and mercury of the population of the EU Member States. Brussels: Directorate-General of Health and Consumer Protection, European Commission.
Shiomi K., 1994. Arsenic in marine organisms: chemical forms and toxicological aspects. In: Nriagu J., ed. Arsenic in the environment. Part II: Human health and ecosystem effects. New York, USA: John Wiley & Sons.
Simon M. & Wuhl-Couturier G., 2002. In: Bohnet F., ed. Ullmann’s encyclopedia of industrial chemistry. 6th ed. Weinheim, Germany: Wiley-VCH.
Sloth J.J., Julshamn K. & Lundebye A.K., 2005a. Total and inorganic arsenic content in Norwegian fish feed products. Aquacult. Nutr., 11(1), 61-66.
Sloth J.J., Larsen E.H. & Julshamn K., 2005b. Survey of inorganic arsenic in marine animals and marine certified reference materials by anion exchange high-performance liquid chromatography-inductively coupled plasma mass spectrometry. J. Agric. Food Chem., 53(15), 6011-6018.
Sloth J.J. & Julshamn K., 2008. Survey of total and inorganic arsenic content in blue mussels (Mytilus edulis L.) from Norwegian fjords: revelation of unusual high levels of inorganic arsenic. J. Agric. Food Chem., 56(4), 1269-1273.
Taleshi M.S. et al., 2010. Arsenic-containing lipids are natural constituents of sashimi tuna. Environ. Sci. Technol., 44, 1478-1483.
Templeton D.M. et al., 2000. Guidelines for terms related to chemical speciation and fractionation of elements. Definitions, structural aspects, and methodological approaches. Pure Appl. Chem., 72(8), 1453-1470.
Uneyama C., Toda M., Yamamoto M. & Morikawa K., 2007. Arsenic in various foods: cumulative data. Food Addit. Contam., 24(5), 447-534.
US EPA, 1998. Integrated Risk Information System, hhtp://www.epa.gov/ncea/iris/subst/0278.htm#quaoral, (04.10.1998).
WHO (World Health Organization), 1988. WHO food additives series No. 24. Roma: FAO/WHO.
WHO (World Health Organization), 2003. Joint FAO/WHO expert committee on food additives. Sixty-first meeting, Roma, 10-19 June 2003. Summary and conclusions. Roma: FAO/WHO.
Yoneda S. & Suzuki K.T., 1997. Equimolar Hg-Se complex binds to selenoprotein P. Biochem. Biophys. Res. Commun., 231(1), 7-11.
Om dit artikel te citeren:
Over : Rikke Vingborg Hedegaard
Technical University of Denmark. National Food Institute. Division of Food Chemistry. Mørkhøj Bygade, 19. DK-2860 Søborg (Denmark). E-mail: jjsl@food.dtu.dk
Over : Jens Jørgen Sloth
Technical University of Denmark. National Food Institute. Division of Food Chemistry. Mørkhøj Bygade, 19. DK-2860 Søborg (Denmark).






