- Startpagina tijdschrift
- volume 13 (2009)
- numéro 2
- Morphological and allozyme variation in a collection of Lagenaria siceraria (Molina) Standl. from Côte d’Ivoire
Weergave(s): 3961 (28 ULiège)
Download(s): 166 (1 ULiège)
Morphological and allozyme variation in a collection of Lagenaria siceraria (Molina) Standl. from Côte d’Ivoire

Nota's van de redactie
Received on February 27, 2008, accepted on September 4, 2008
Résumé
Variation morphologique et enzymatique dans une collection de Lagenaria siceraria (Molina) Standl. en Côte d’Ivoire. Cette étude décrit la variabilité intraspécifique de 30 accessions de Lagenaria siceraria à graines consommées de la collection de l’Université d’Abobo-Adjamé. Ces accessions proviennent de trois zones géographiques de la Côte d’Ivoire (Centre, Est et Sud). La sélection opérée par les paysans sur la base de la taille des graines subdivise cette espèce en deux cultivars : le cultivar à petites graines et le cultivar à grosses graines. Les études morphologiques impliquent 18 accessions et 24 caractères morphologiques. L’analyse multivariée de variance (MANOVA) a montré une différence significative entre les deux cultivars. L’analyse en composantes principales portant sur 13 caractères morphologiques a révélé une variation entre les individus analysés, principalement sur base de la taille des fleurs, fruits et graines. Le dendrogramme construit avec la méthode UPGMA a permis un regroupement des cultivars. L’analyse de la structure génétique basée sur les marqueurs allozymiques a donné les valeurs suivantes : 18,95 % pour le pourcentage de loci polymorphes (P), 1,21 pour le nombre moyen d'allèles (A) et 0,053 pour l’hétérozygotie observée (Ho). La diversité génétique intra-accession (HS = 0,188) est plus élevée que la diversité génétique inter-accessions (DST = 0,082). Les estimations des F-statistiques indiquent un faible niveau de différentiation génétique entre les accessions (FST = 0,298), suggérant seulement 30 % de variation entre les accessions. La distance génétique de Nei entre les deux cultivars est également faible (0,002), indiquant que les deux cultivars sont génétiquement similaires et pourraient appartenir au même groupe génétique.
Abstract
This study describes the intraspecific variation of 30 edible-seed Lagenaria siceraria germplasm accessions from the University of Abobo-Adjamé. These accessions were collected from three (Centre, East and South) geographical zones of Côte d’Ivoire. Selection based on seed size by the farmers has resulted in subdividing the species into two cultivars: large-seeded and small-seeded. The morphological diversity study of the collection included 18 accessions and 24 traits. The multivariate analysis of variance (MANOVA) showed a significant difference between the two groups of cultivars. Principal component analysis on 13 traits pointed out variations among individuals, mainly on the basis of flower, fruit, and seed size. Dendrogram with UPGMA method allowed clustering of the cultivars. The genetic structure analysis among accessions using allozyme markers showed the following values: 18.95% for the proportion of polymorphic loci (P), 1.21 for the number of alleles (A) and 0.053 for observed heterozygosity (Ho). The level of the within accessions genetic diversity (HS = 0.188) was higher than the genetic variation among accessions (DST = 0.082). The estimates of F-statistics indicated a low level of genetic differentiation between accessions (FST = 0.298). Such a value suggested that L. siceraria maintains about 30% of its genetic variation among accessions. Nei genetic distances between the two cultivars were also low (0.002), indicating that cultivars were genetically similar enough to belong to the same genetic group.
Inhoudstafel
1. Introduction
1Lagenaria siceraria (Molina) Standl. is an important crop in tropical Africa. Some cultivars of this species with others of the Cucurbitaceae family are called Egusi in Benin, Nigeria, and pistachio in Côte d’Ivoire. Although these crops are neglected and underutilized in Africa, their cultivation is widespread in most West African countries (Achu et al., 2005; Achigan-Dako et al., 2006; Zoro Bi et al., 2006).
2These cucurbits are prized for their oilseeds and are consumed as soup thickener. Their seeds are good sources of lipids and proteins (Achu et al., 2005; Loukou et al., 2007). Like other neglected and underutilized crops in Africa, cucurbits have numerous agronomic and economic potentials. They are well adapted to extremely divergent agroecosystems and to various cropping systems. They are also characterized by minimal inputs. Increased production and use of these cucurbits can result in food security and diversify small farmer’s income (Chweya et al., 1999; Williams et al., 2002).
3In developing countries, the exploitation of local resources is certainly a way to achieve the objective of food security, particularly for a fast-growing population. However this requires the preservation and availability of a high level of genetic diversity of these resources (Given, 1987). This conservation constitutes a challenge for crops such as cucurbits that are endangered and have been neglected by national research programs. To this regard prerequisite information on genetic pattern is needed to make appropriate decision.
4In spite of the economic importance of edible seeds of L. siceraria in Côte d’Ivoire, knowledge of its genetic diversity and differentiation is very poor. In order to fill up this gap, it is crucial to collect the genetic resource available at country level and to characterize the genetic diversity of the collected accessions on the basis of several markers.
5The objective of this study is to characterize the collection of L. siceraria from different geographical zones in Côte d’Ivoire and available at Abobo-Adjamé University. The variability will be assessed among accessions and cultivars, using in particular morphological and enzyme markers. In our investigations, we decided to use biochemical markers, such as allozymes, because they are relevant to identify the heterozygosity and to better know the genetic structure of the species. Results obtained from this characterization would contribute to sample fruits and seeds in the most representative areas, to conduct further molecular analysis and to define appropriate sampling strategies for the conservation of L. siceraria genetic resources in Côte d’Ivoire.
2. Material and methods
2.1. Plant material and collection sites
6Thirty accessions (Table 1) of L. siceraria were selected from the germplasm collection maintained at the University of Abobo-Adjamé (Abidjan, Côte d’Ivoire). An accession is a sample collected in one field or obtained from one farmer’s stock. The selected accessions were identified by alpha-numeric codes. A minimum distance of 25 km separated two consecutive collecting sites in each zone. The seed samples were collected mainly in three geographical zones (South, East, and Centre) of Côte d’Ivoire. The geographical coordinates and ecological traits of sites of the collecting missions are as follows (Zoro Bi et al., 2005):
7– The southern zone which is localized between latitudes 4°41 N-6°00 N and longitudes 4°00 W-7°30 W. In this zone, rainfalls are abundant (annual mean > 2,000 mm) and mean annual temperature is 28°C, with annual amplitude of 5-10°C. Vegetation is mainly represented by the tropical rain forest, with mangrove on the coastal side.
8- The eastern zone which is limited by latitudes 6°00 N-8°00 N and longitudes 3°00 W-5°00 W. This zone is characterized by the transitional woodland savannas, with several blocks of semi-deciduous forests. Rainfalls vary from 875 to 1,910 mm, with an annual mean of 1,250 mm; the annual mean temperature is 27°C.
9- The central zone which is limited by latitudes 6°00 N-8°00 N and longitudes 5°00 W-7°00 W. Annual rainfalls vary from 800 to 1,400 mm, with an annual mean of 1,200 mm; the annual mean temperature is 27°C. The vegetations are made of various woodland savannas with extended ranges of herbaceous areas.
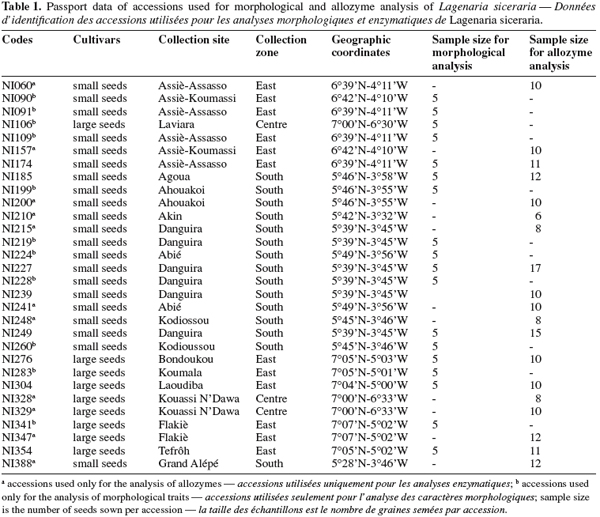
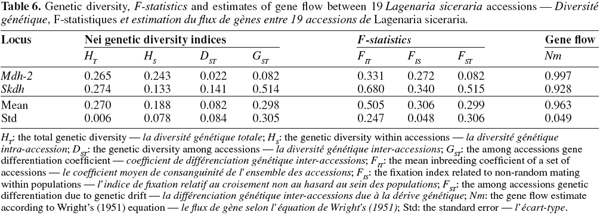
10Accessions used in this study are from two landraces known as small-seed and large-seed. For the morphological study 18 accessions with 12 small-seeded and 6 large-seeded accessions were used. For allozyme analysis, 13 small-seeded and 6 large-seeded accessions were used. The number of seeds used per accession for both morphological and allozyme analysis are indicated in table 1.
2.2. Morphological characterization
11Study site and experimental design. The study was carried out in Abidjan district from May to November 2007. The experimental site was located in Akouedo village in Abidjan suburb, between latitudes 5°17’N-5°31’N and longitudes 3°45’W-4°22’W. In this zone, rainfalls are abundant (annual mean > 2,000 mm) and the mean temperature is 28°C, with annual amplitude of 5-10°C. The field lay out was a completely randomized design, with three replications. Each replicate consisted of a 30 m x 27 m plot containing 90 plants (i.e., the eighteen accessions), each accession being represented by 5 plants. The planting distance was 3 m between and within rows with 1.5 m of edges. Two consecutive plots were spaced by 3 m. Manual weeding was carried out during plant development.
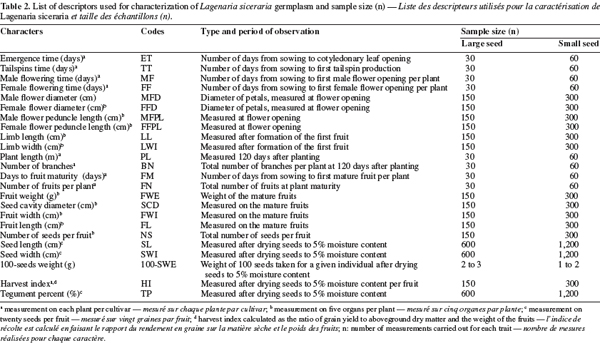
12Morphological data and analysis. Morphological diversity was characterized using standard descriptors for cucurbits: 24 characters were chosen among those published for Citrullus lanatus (Thunb.) Matsum. & Nakai (Maggs-Kölling et al., 2000) and Lagenaria siceraria (Morimoto et al., 2005) (Table 2). Multivariate analysis of variance (MANOVA) was performed with SAS software package (SAS, 1999) to investigate the difference between the two cultivars. Principal Components Analysis (PCA) with Statistica software package (Statistica, 1995) was applied to further describe morphological variation among accessions and between cultivars. PCA is particularly relevant to describe dataset by combining correlated variables into factors. Prior to PCA, the average values of the traits were standardized according to the formula:

13This standardization is required to reach the same scale for all the characters (Dagnelie, 1998). Data were averaged across individuals for each accession, and UPGMA (Unweighted Pair Group Method with Arithmetic) dendrograms were computed by Statistica software package (Statistica, 1995) to describe the relationships between accessions. To determine morphological differences among accessions from the three zones of the collecting missions, multivariate analysis of variance was carried out.
2.3. Allozyme analysis
14Electrophoresis. Isozyme assays were conducted on cotyledonary tissue. The selected seeds were sown in field and cotyledons were taken from each 3-4 days old seedling. A quantity of 0.01 g of cotyledonary tissue from each seedling was ground in 0.045M TRIS-HCl, pH 7.1 (Knerr et al., 1989; Staub et al., 1997). Plant tissue was held at -20°C until the horizontal starch gel electrophoresis was performed according to Zoro Bi (1999).
15Gels were prepared using 60 g of hydrolyzed potato starch from Sigma (Sigma # S-5651) and 15 g of sucrose that were dissolved in 600 ml of buffer. The continuous morpholine-citrate, pH 6.1 was employed for electrophoresis.
16Four enzyme systems were used to study electrophoretic variation: alcohol dehydrogenase (ADH, E.C. 1.1.1.1), malate dehydrogenase (MDH, E.C. 1.1.1.37), malic enzyme (ME, E.C. 1.11.1.7), shikimate dehydrogenase (SKDH, E.C. 1.1.1.25). The techniques for histochemical staining procedures were those reported by Zoro Bi (1999) with Lima bean (Phaseolus lunatus L.). For each enzymatic system, the presumed loci were numbered in ascending order from the anode. For each isozyme, the most common allele was referred to as 100 and the other alleles were named according to their migration distance in millimetres using the standard (Koenig et al., 1989).
17Data analysis. Allozyme data analysis was performed using the computer program GENSURVEY (Vekemans et al., 1997). Statistics of genetic diversity within and among accessions were calculated: percentage of polymorphic loci at 5% level (P), mean number of alleles per locus (A), observed heterozygosity (Ho), genetic diversity (He) corrected for small sample size (Nei, 1978) and Wright’s F [F = (1-Ho/He)], the inbreeding coefficient which measures the deviation of the population genotypic composition from Hardy-Weinberg (H-W) expectations. Deviation from Hardy-Weinberg equilibrium at each locus in each accession and heterogeneity in alleles frequencies among accessions were tested by chi-square (χ2) using the computer program GENEPOP (Raymond et al., 1995). Genetic structure of the accessions was investigated using Nei’s genetic diversity analysis on polymorphic loci (Nei, 1973). The genetic differentiation among accessions and cultivars was also estimated by partitioning the total genetic diversity (HT) into gene diversity within accessions or cultivars (HS) and among accessions or cultivars (DST) i.e. HT = HS + DST. The degree of genetic differentiation (GST) was calculated as DST/HT. Wright’s fixation index within population (FIS), among populations (FST) and total genetic differentiation (FIT) were calculated to demonstrate the relative distribution of genetic variation among and within accessions and cultivars (Wright, 1965). The number of migrants into accessions per generation (Nm) was estimated (Wright, 1951). Cultivars divergence was estimated using Nei’s genetic distance (Nei, 1978).
3. Results
3.1. Morphological variation
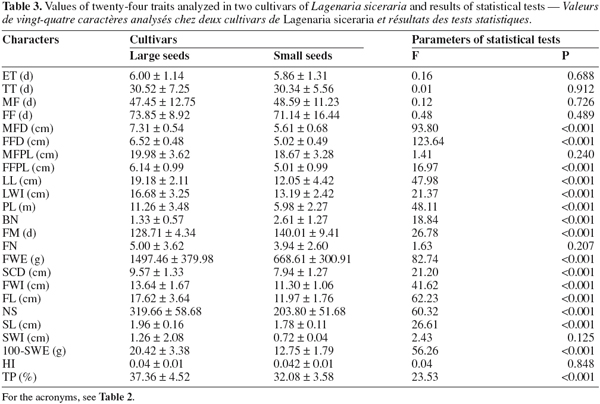
18Comparison of morphological traits using a multivariate analysis of variance (MANOVA) showed a significant difference between the two cultivars of L. siceraria (F = 45.21; P < 0.001). As shown in table 3, this difference is based on 16 traits: male flower diameter (MFD), female flower diameter (FFD), female flower peduncle length (FFLP), limb length (LL), limb width (LWI), plant length (PL), number of branches (BN), days to fruit maturity (FM), fruit weight (FWE), seed cavity diameter (SCD), fruit width (FWI), fruit length (FL), number of seed per fruit (NS), seed length (SL), 100-seeds weight (100-SWE), tegument percent (TP). The large-seeded cultivar gave the highest values for all these traits with the exception of the number of branches from the central taproot (BN) and fruit maturity (FM).
19A minimum list of descriptors was selected from the establishment of a correlations matrix (Table 4). When we observed a high positive correlation (> 0.70) between two variables, only one was retained to avoid redundancy (Table 4). Consequently 13 variables: seed emergence time (ET), days to the first tendril appearance (TT), days to the first female flower (FF), male flower diameter (MFD), female flower peduncle length (FFPL), days to the first fruit maturity (FM), fruit width (FWI), number of seeds per fruit (NS), seed length (SL), seed width (SWI), 100-seeds weight (100-SWE), harvest index (HI), and tegument percent (TP) were used for the morphological characterization of the accessions.
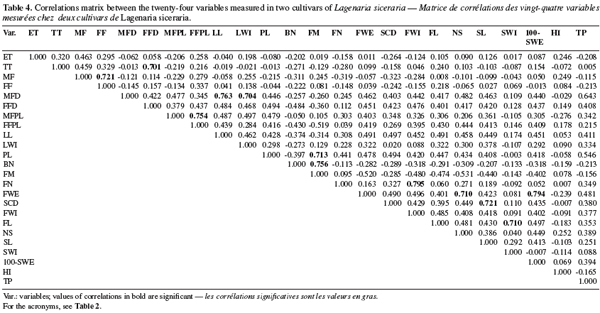
20The first two principal components accounted for 46.398% of the total variability (eigenvalues > 1). Male flower diameters (MFD), fruit width (FWI), number of seeds (SN), 100-seeds weight (100-SWE) are correlated with the first component (representing 32.762 % of the total variability). These variables are negatively correlated with PC1. The second component (13.636% of the variability) is mainly linked to the seed emergence times (ET) with a positive correlation. Figure 1 shows the correlation circle of the two first principal components analysis.
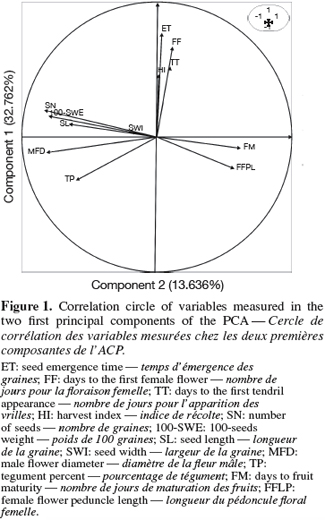
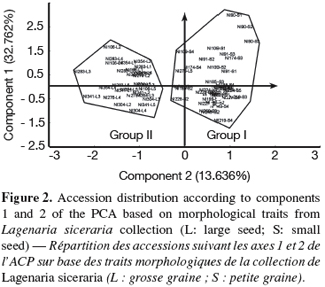
21On the basis of these traits, two morphological groups were formed (Figure 2). The group 1 included individuals from the small-seeded cultivar, characterized by late maturity and small size of female flower peduncle. The group 2 consisted of individuals of large-seeded cultivar, characterized by large diameter of petals opening, and heavy fruits of large sections with many seeds per fruit.
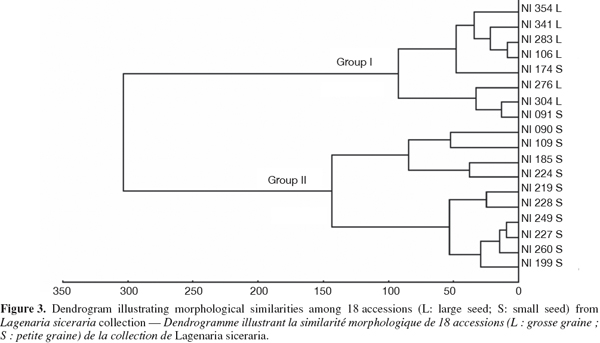
22UPGMA cluster analysis of morphological differentiation among accessions computed from a matrix of pairwise Euclidean distances showed two distinct groups (Figure 3). Group 1 included all accessions belonging to the large-seeded cultivar and two accessions (NI174, NI091) from the small-seeded cultivar. Group 2 included only accessions from the small-seeded cultivar. Consequently, dendrogram analysis showed that the two cultivars were morphologically dissimilar since only two accessions (11%) were inappropriately clustered.
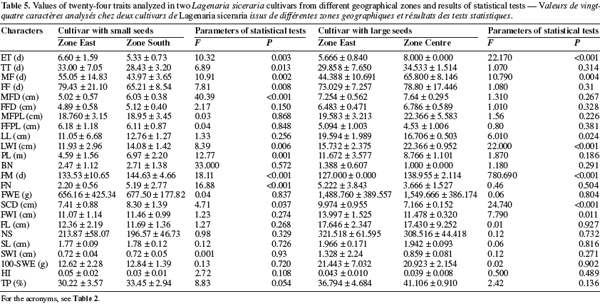
23The multivariate analysis of variance (MANOVA) applied to the small-seeded cultivar obtained from the South and East showed significant differences between the two zones (F = 23.25; P < .001). This result is mainly due to 10 characters out of 24 measured: emergence time (ET), tailspins time (TT), male flowering time (MF), female flowering time (FF), male flower diameter (MFD), limb width (LWI), plant length (PL), days to fruit maturity (FM), number of fruits per plant (FN), seed cavity diameter (SCD) (Table 5). For the large-seeded cultivar obtained from the Centre and the East, the multivariate analysis also showed a significant difference between the two areas (F = 21.88; P < 0.001). This difference is due to 7 characters out of the 24 measured: emergence time (ET), male flowering time (MF), limb length (LL), limb width (LWI), days to fruit maturity (FM), seed cavity diameter (SCD), fruit width (FWI) (Table 5). These data suggest that the morphological differences between accessions belonging to the same cultivar but collected in distinct geographical zones are not important.
3.2. Allozymes variation and genetic structure
24Among the four enzyme systems investigated, Adh revealed three loci with a total of four alleles, ME showed only one locus with one allele, Mdh showed two loci with three alleles and Skdh revealed one locus with two alleles. Two loci (Adh-2 and ME) produced unclear patterns and were discarded. Thus five loci were taken into account for further analysis. Two loci were polymorphic and diallelic: Mdh-2 and Skdh, whereas Mdh-1, Adh-1 and Adh-3 were monomorphic.
25The proportion of polymorphic loci per accession (P) varied from 0 (e.g., NI060) to 40% (e.g., NI388) with a mean of 18.95%. The mean number of alleles (A) per locus varied from 1.0 (e.g., NI210) to 1.4 (e.g., NI248) with an average of 1.2, indicating a low allelic richness. A low genetic diversity was also observed. Indeed, the average observed heterozygosity was 0.053 ranging from 0 (e.g., NI249) to 1.4 (e.g., NI241) and the average expected heterozygosity (He) was 0.073 ranging from 0 (e.g., NI329) to 0.202 (e.g., NI276). The observed mean heterozygosity (Ho = 0.053) was similar to the average expected heterozygosity. This suggested that the populations were at Hardy-Weinberg equilibrium. According to the Chi-square test, 66.67% of the accessions were not significantly different from zero (α = 0.05) confirming the Hardy-Weinberg equilibrium hypothesis. This indicated that most accessions did not deviate from random union of gametes.
26The total gene diversity (HT) was 0.270 (Table 6). For Mdh-2 locus, the largest proportion of diversity was attributable to the within-accessions component (HS = 0.243; DST = 0.022). However, for Skdh locus, difference between the within-accessions and the among-accessions components of diversity (HS = 0.133; DST = 0.141) was not significant. On the other hand, the coefficient of gene differentiation (GST) between accessions was estimated at 29.8%, indicating that most of the total genetic diversity in this species was within accessions rather than among accessions. From table 6, the mean inbreeding index (FIT) for the 19 accessions was 0.505. This relatively high value showed important deficiency in heterozygosity. The average FIS of 0.306 indicated a slight deficit of heterozygotes within accessions. On the other hand, the average FST of 0.299 showed a low genetic differentiation among accessions. The mean number of migrants per generation (Nm) based on Wright’s equation was 0.963. Such a value indicated that on average, one individual migrated in a given accession (seed stock or field) per generation.
27At the cultivar level (Table 7), the average of total gene diversity (HT) and intra-cultivar genetic diversity (HS) were 0.300 and 0.298, respectively. The inter-cultivar genetic diversity (DST) and the coefficient of gene differentiation among cultivars (GST) were 0.002 and 0.007 respectively. According to these results, the degree of genetic differentiation between cultivars was very low (only 0.7%). F-statistics for the two cultivars indicated a relatively high mean inbreeding index (FIT = 0.569), showing an important deficiency in heterozygosity. A high value was also obtained for FIS (0.567). The proportion of the total genetic diversity among cultivars (FST = 0.006) was very low. A low value was also estimated for Nei genetic distances between the two cultivars, with D = 0.002, indicating that cultivars were genetically similar enough to belong to the same genetic group.

4. Discussion
4.1. Morphological variation
28Our study showed morphological heterogeneity within the collection of L. siceraria of Abobo-Adjamé University in Côte d’Ivoire. According to the multivariate analysis of variance, difference between the two groups of accessions with small and large seeds size was significant. Fruit and seed shape and size are reported to be highly variable in the genus Lagenaria (Bisognin, 2002). In most rural African communities, the landraces of L. siceraria are generally distinguished by their fruit size and shape, and designated by common name according to these morphological differences (Morimoto et al., 2005). In Côte d’Ivoire, farmers refer to seed size when designating cultivars of edible-seeded L. siceraria (Zoro Bi et al., 2006). Also, in the cucurbit family, the significant contribution of fruit and seed traits to morphological variability has been reported for watermelon (Maggs-Kölling et al., 2000; Gusmini, 2003), bottle gourd (Morimoto et al., 2005) and squash (Paris, 2001).
29However, in estimating the relative contribution of the different traits in the overall phenotypic variation among the 18 accessions, the first two principal components (PCs) explained 46.398% of the diversity obtained from the 13 selected traits. The variability explained by the first PC alone (32.762%) was mainly due to variations in fruit (FWI), seed (NS, 100-SWE) and flower sizes (MFD). In the cucurbit family, the significant contribution of fruit and seed traits to morphological variability has been reported for watermelon (Maggs-Kölling et al., 2000; Gusmini, 2003). In spite of a variability explained by the first two principal components inferior to 50%, a relative separation was observed between the two cultivars (Figure 2). The presence of one individual (NI276-L5) from the large seeded accession (NI276) in the group 1 made up of small-seeded accessions could result from accidental mixture by farmers or collectors. However Morimoto et al. (2005) reported the difficulty to classify the landraces of L. siceraria into distinct groups, because of the high and continuous morphological variation in the species. The difference between the two studies results from the fact that we analyze only two edible-seed cultivars while Morimoto et al. (2005) analyzed several forms of L. siceraria and its wild relatives.
30A strong positive correlation between the weight of the fruits and the number of seeds was observed in our study. The same positive correlation was also noted in watermelon (Nerson, 2002). Therefore the fruit weight of L. siceraria could be used as a good criteria to select individuals with higher number of seeds. This result is congruent with findings of Achigan-Dako et al. (2006).
31The phenotypical variation between the two cultivars of L. siceraria from Côte d’Ivoire seemed to be uncorrelated to the different regions where samples were collected. Two hypotheses could explain such a result:
32– the occurrence of an important seed flow,
33– the similarity of the cultivation history among the collecting zones.
34According to Montes-Hernandez et al. (2002), human activities significantly buffer genetic variability between plants occurring in different geographical regions. This argument could be supported by the fact that most often farmers exchange seeds among themselves. Fruits and seeds are selected to constitute planting material for the next season. In addition, parts of the harvested products are marketed and contribute to the movement of seeds between regions.
4.2. Genetic diversity
35In general, the intra-accession polymorphism indices estimated in this study were low (P = 18.95%; A = 1.21; He = 0.073). These indices are similar to those reported by Decker-Walters et al. (1990) on Cucurbita maxima Duchesne (P = 11.5%; A = 1.43; He = 0.039) and on Cucurbita pepo L. (P = 19.3%; A = 2.24; He = 0.068). However values from L. siceraria were smaller than those calculated in four Cucurbita taxa by Montes-Hernandez et al. (2002): P = 100%; A = 2.08; He = 0.407 for Cucurbita argyrosperma ssp. sororia Huber, P = 93%; A = 2.5; He = 0.391 for Cucurbita argyrosperma ssp. Argyrosperma Hort Huber, P = 97%; A = 2.06; He = 0.496 for Cucurbita moschata Duchesne and P = 92%; A = 2.08; He = 0.366 for C. pepo. The lack of RAPD diversity (known to be highly polymorphic) was also found in southern African landraces of L. siceraria (Decker-Walters et al., 2001) and in several accessions of two Citrullus species: Citrullus lanatus and Citrullus colocynthis (L.) Schrad. (Levi et al., 2001). These results confirm our study and pointed out a narrow genetic basis in this species.
36In spite of the very low number of loci and individuals analyzed in this study, two hypotheses could explain the low allelic richness: the selection favouring homozygote individuals and the founder effect. However, the reproductive biology of indigeneous cucurbit avoiding selfing makes the first hypothesis (homozygotes selection) improbable. Indeed, L. siceraria is monoecious (staminate and pistilate flowers are separated) and flowers are pollinated by various insects. Thus this species is bound to experiment insect-mediated cross pollination which promotes random-mating, buffering homozygotes selection (Wright, 1951). This argument was supported by the high percentage of accessions (66.67%) not significantly different from Hardy-Weinberg equilibrium. The last hypothesis: the founder effect, due to farmer’s seeds selection approach, is most likely the major cause of the low allelic richness. In particular, in the collecting sites, generally a low number of seeds is usually taken from the farmer’s stock, or are obtained from local markets, resulting to the genetic variability depletion (Nei et al., 1975).
37In our study, trends of variation were not similar with the application of the two markers: morphology and allozymes. Differentiation among cultivars was much higher for morphological traits than for allozymes. Djè et al. (1998) found similar results for sorghum landraces of northwestern Morocco. During the selection process, farmers and breeders favour phenotypic diversity, in order to meet varietal adaptation to diverse cropping systems and consumer’s requirements. Considering the low genetic variability observed in our L. siceraria collection, one can assume that allozyme markers were not powerful enough to capture the genetic basis of the morphological variation, probably due to complex and multigenic inheritance of fruit and seed traits in cucurbits (Guner et al., 2004).
4.3. Genetic structure and gene flow
38The mean within-accession gene diversity index (HS = 0.188) was similar to those reported by Hamrick et al. (1997) for cross-pollinated plants. Indeed, these authors showed that intra-population gene diversity index varied between 0.103 and 0.266. The degree of genetic differentiation between accessions (GST = 0.298) was also similar to those reported for many cross-pollinated plants, GST = 0.234 (Hamrick et al., 1997). The low values of GST in the present study could result from the high level of gene flow among accessions, in particular the frequent seed exchange among farmers. However, our GST value was lower than the value estimated in Schizopepon bryoniaefolius Maxim. (GST = 0.68), a wild Cucurbitaceae (Akimoto et al., 1999). Differences between our study and Akimoto et al. (1999) study could be attributed to the different floral biology between the two species. L. siceraria is monoecious (male and female flowers are separated) and predisposed to predominantly outcrossing, while S. bryoniaefolius is androdioecious (with hermaphrodite flowers) which favours selfcrossing.
39On average, FIS showed a significant deficiency of heterozygosity (FIS > 0) for all accessions. The proportion of the total genetic diversity found among accessions (FST = 0.299) was higher than that described by Hamrick et al. (1989) in animal-pollinated species (FST = 0.187), and almost similar to the larger group including the cross-pollinated plants (FST = 0.234). The gene flow estimation (Nm = 0.963) and the low genetic differentiation between accessions (FST = 0.299) confirmed the high rate of gene exchange between accessions.
5. Conclusion
40This study has allowed a better knowledge of the cultivated L. siceraria collection of the University of Abobo-Adjamé (Abidjan, Côte d’Ivoire). Morphological characterization showed significant difference between two cultivars: small-seeded and large-seeded cultivars. Isozyme electrophoresis data indicated a lower genetic heterogeneity among accessions than within accessions. Therefore, during the collecting missions, it is recommended to collect a sufficient number of seeds or fruits within each accession, which is better than attempting to collect as many accessions as possible. However the low number of analyzed loci and individuals suggest that analysis of additional accessions is required before a definitive conclusion can be performed. On the other hand, many questions still remain to be solved. An in-depth knowledge of reproductive biology of this species and the use of DNA markers are required. Indeed, molecular markers can be an effective means to determine genetic relatedness among cultivars and among accessions present in L. siceraria germplasm collection. These markers are generally more polymorphic than isozymes. Results obtained in studying floral biology and molecular variability will help us to define sampling strategies and optimum sample size for the management of L. siceraria genetic resources.
41Acknowledgements
42This research was financed by the « Direction Générale de la Coopération au Développement » (DGCD, Brussels, Belgium) and supervised by the « Commission Universitaire pour le Développement » (CUD, Brussels, Belgium).
Bibliographie
Achigan-Dako E.G. et al., 2006. Evaluation agronomique de trois espèces de Egusi (Cucurbitaceae) utilisées dans l'alimentation au Bénin et élaboration d'un modèle de prédiction du rendement. Biotechnol. Agron. Soc. Environ., 10(2), 121-129.
Achu B.M. et al., 2005. Nutritive value of some Cucurbitaceae oilseeds from different regions in Cameroon. Afr. J. Biotechnol., 4(11), 1329-1334.
Akimoto J., Fukuhara T. & Kikuzawa K., 1999. Sex ratios and genetic variation in a functionally androdioecious species, Schizopepon bryoniaefolius (Cucurbitaceae). Am. J. Bot., 86(6), 880-886.
Bisognin D.A., 2002. Origin and evolution of cultivated cucurbits. Cienc. Rural, 32, 715-723.
Chweya J.A. & Eyzaguirre P.B., 1999. The biodiversity of traditional leavy vegetables. Roma: IPGRI.
Dagnelie P., 1998. Statistique théorique et appliquée. Bruxelles : De Boeck & Larcier.
Decker-Walters S.D., Walters T.W., Posluszny U. & Kevan P.G., 1990. Genealogy and gene flow among annual domesticated species of Cucurbita. Can. J. Bot., 68, 782-789.
Decker-Walters D., Staub J., Lopez-Sese A. & Nakata E., 2001. Diversity in landraces and cultivars of bottle gourd (Lagenaria siceraria; Cucurbitaceae) as assessed by Random Amplified Polymorphic DNA. Genet. Resour. Crop Evol., 48, 369-380.
Djè Y., Ater M., Lefèbvre C. & Vekemans X., 1998. Patterns of morphological and allozyme variation in sorghum landraces of Northwestern Morocco. Genet. Resour. Crop Evol., 45, 541-548.
Given D.R., 1987. What the conservationist requires of ex situ collections. In: Branwell D., Hamann O., Heywood V. & Synge H., eds. Botanic gardens and the world conservation strategy. London: Academic Press, 103-116.
Guner N. & Whener T.C., 2004. The genes of watermelon. Hortic. Sci., 39(6), 1175-1182.
Gusmini G., 2003. Watermelon (Citrullus lanatus) breeding handbook. Raleigh, NC, USA : North Carolina State University.
Hamrick J.L. & Godt M.J.W., 1989. Allozyme diversity in plant species. In: Brown A.H.D., Clegg M.T., Kahler A.M. & Weir B.S. Plant population genetics, breeding, and genetic resources. Sunderland, Massachusetts, USA: Sinauer Associates, 43-63.
Hamrick J.L. & Godt M.J.W., 1997. Allozyme diversity in cultivated crops. Crop Sci., 37, 26-30.
Knerr L.D., Staub J.E., Holder D.J. & May B.P., 1989. Genetic diversity in Cucumis sativus L. assessed by variation at 18 allozyme coding loci. Theor. Appl. Genet., 78, 119-128.
Koenig R. & Gepts P., 1989. Allozyme diversity in wild Phaseolus vulgaris: further evidence for two major centers of genetic diversity. Theor. Appl. Genet., 78, 455-459.
Levi A., Thomas C.E., Wehner T.C. & Zhang X., 2001. Low genetic diversity indicates the need to broaden the genetic base of cultivated watermelon. Hortic. Sci., 36(6), 1096-1101.
Loukou A.L. et al., 2007. Macronutrient composition of three cucurbit species cultivated for seed consumption in Côte d'Ivoire. Afr. J. Biotechnol., 6(5), 529-533.
Maggs-Kölling G., Madsen S. & Christiansen J., 2000. A phenetic analysis of morphological variation in Citrullus lanatus in Namibia. Genet. Resour. Crop Evol., 47, 385-393.
Montes-Hernandez S. & Eguiarte L.E., 2002. Genetic structure and indirect estimates of gene flow in three taxa of Cucurbita (Cucurbitaceae) in western Mexico. Am. J. Bot., 89(7), 1156-1163.
Morimoto Y., Maundu P., Fujimaki H. & Morishima H., 2005. Diversity of landraces of the white-flowered gourd (Lagenaria siceraria) and its wild relatives in Kenya: fruit and seed morphology. Genet. Resour. Crop Evol., 52, 737-747.
Nei M., 1973. Analysis of gene diversity in subdivided populations. Proc. Natl Acad. Sci. USA, 70(12), 3321-3323.
Nei M., Maruyama T. & Chakraborty R., 1975. The bottleneck effect and genetic variability in populations. Evolution, 29, 1-10.
Nei M., 1978. Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics, 89, 583-590.
Nerson H., 2002. Relationships between plant density and fruit and seed production in muskmelon. J. Am. Soc. Hortic. Sci., 127(5), 245-256.
Paris H.S., 2001. Characterization of Cucurbita pepo collection at the Newe Ya'ar Research Center, Israël. Plant Genet. Resour. Newsl., 126, 41-45.
Raymond M. & Rousset F., 1995. Genepop (version 1.2): population genetics software for exact tests and ecumenicism. J. Hered., 86, 248-249.
SAS, 1999. SAS/ETS User's Guide, version 6. 4th ed. Cary, NC, USA: SAS Institute.
Statistica, 1995. Statistica for windows: release 7.0. Tulsa, OK, USA: StatSoft Inc.
Staub J.E., Serquen F.C. & McCreight J.D., 1997. Genetic diversity in cucumber (Cucumis sativus L.): III. An evaluation of Indian germplasm. Genet. Resour. Crop Evol., 44, 315-326.
Vekemans X. & Lefèbvre C., 1997. On the evolution of heavy-metal tolerant populations in America maritima: evidence from allozyme variation and reproductive barriers. J. Evol. Biol., 10, 175-191.
Williams J.T. & Haq N., 2002. Global research on underutilized crops. An assessment of current activities and proposal for enhanced cooperation. Southampton, UK: ICUC.
Wright S., 1951. The genetical structure of populations. Ann. Eugen., 15, 6531-6535.
Wright S., 1965. The interpretation of population structure by F-statistics with special regard to systems of mating. Evolution, 19, 395-420.
Zoro Bi I.A., 1999. Variabilité génétique des populations sauvages de Phaseolus lunatus L. dans la vallée centrale du Costa Rica et ses implications dans la mise au point d'une stratégie de conservation in situ. Thèse de doctorat : Faculté universitaire des Sciences agronomiques de Gembloux (Belgique).
Zoro Bi I.A. et al., 2005. Biodiversity of cucurbits consumed as sauce thickener in Ivory Coast: a capital resource for the economic prosperity of rural women. In: Segers H., Desmet P. & Baus E., eds. Tropical biodiversity: science, data, conservation. Proceedings of the 3rd GBIF science symposium, 18-19 April 2005, Brussels, 158-167.
Zoro Bi I.A. et al., 2006. Indigenous cucurbit of Côte d'Ivoire: a review of their genetic resources. Sci. Nat., 3(1), 1-9.
Om dit artikel te citeren:
Over : Kévin K. Koffi
Université d'Abobo-Adjamé. Unité de Formation et de Recherche des Sciences de la Nature. 02 BP 801. CI-Abidjan 02 (Côte d'Ivoire).
Over : Guy K. Anzara
Université d'Abobo-Adjamé. Unité de Formation et de Recherche des Sciences de la Nature. 02 BP 801. CI-Abidjan 02 (Côte d'Ivoire).
Over : Marie Malice
Gembloux Agricultural University – FUSAGx. Unité de Phytotechnie tropicale et Horticulture. Passage des Déportés, 2. B-5030 Gembloux (Belgium).
Over : Yao Djè
Université d'Abobo-Adjamé. Unité de Formation et de Recherche des Sciences de la Nature. 02 BP 801. CI-Abidjan 02 (Côte d'Ivoire).
Over : Pierre Bertin
Université catholique de Louvain. Unité d’Ecophysiologie et Amélioration végétale (ECAV). Croix du Sud, 2/11. B-1348 Louvain-la-Neuve (Belgium).
Over : Jean-Pierre Baudoin
Gembloux Agricultural University – FUSAGx. Unité de Phytotechnie tropicale et Horticulture. Passage des Déportés, 2. B-5030 Gembloux (Belgium).
Over : Irié A. Zoro Bi
Université d'Abobo-Adjamé. Unité de Formation et de Recherche des Sciences de la Nature. 02 BP 801. CI-Abidjan 02 (Côte d'Ivoire). E-mail: banhiakalou@yahoo.fr






