- Home
- Volume 30 (2026)
- Numéro 2
- Floristic characterisation and population status of Erythrina senegalensis DC. and Erythrina sigmoidea Hua in the Guiriko region (Burkina Faso)
View(s): 0 (0 ULiège)
Download(s): 0 (0 ULiège)
Floristic characterisation and population status of Erythrina senegalensis DC. and Erythrina sigmoidea Hua in the Guiriko region (Burkina Faso)

Attached document(s)
original pdf fileRésumé
Caractérisation floristique et états des populations d’Erythrina senegalensis DC. et d’Erythrina sigmoidea Hua dans la région du Guiriko (Burkina Faso)
Description du sujet. La dépendance des populations vis-à-vis des formations naturelles affecte la survie des espèces végétales.
Objectifs. Les objectifs sont de caractériser la flore des formations végétales à Erythrina senegalensis et Erythrina sigmoidea dans la région du Guiriko et d’analyser l’état actuel de leurs populations en vue d’orienter leur conservation.
Méthode. Les données floristiques et dendrométriques ont été collectées suivant un échantillonnage orienté à la suite d’une prospection des formations végétales à Erythrina. Les formations végétales retenues pour l’étude ont été les forêts classées de Péni, Dindéresso, Kuinima, Kou et les bois sacrés des collines gréseuses de Kôrô.
Résultats. Une diversité floristique faiblement similaire (Cs = 0,45) avec 213 et 165 espèces respectivement dans la formation à E. senegalensis et dans la formation à E. sigmoidea a été observée avec les Rubiaceae, les Combretaceae, les Fabaceae-ceasalpinioideae, les Anacardiaceae, les Fabaceae-faboideae et les Fabaceae-mimosoideae comme familles dominantes. Les types biologiques les plus dominants sont les Phanérophytes. Une dynamique en régression des populations avec une quasi-absence d’individus de gros diamètres et des densités d’individus adultes de 25,48 individus·ha-1 pour E. senegalensis et 13,15 individus·ha-1 pour E. sigmoidea a été obtenue. Les faibles densités des juvéniles (<< 50 individus·ha-1) et les faibles potentiels de renouvellement (< 2 individus·ha-1) observés constituent un risque majeur pour la survie des populations d’Erythrina. Les populations de E. senegalensis sont plus sujettes à l’écorçage (13,30 % des individus) tandis que celles de E. sigmoidea présentent des impacts de feux de brousse (88 % des individus).
Conclusions. Le renouvellement de ces espèces est gravement compromis dans la région du Guiriko. Des études doivent être menées sur leur mode de régénération et des mesures de protection doivent être prises de concert avec les populations pour leur conservation et gestion durable.
Abstract
Description of the subject. The dependence of populations on natural formations affects the survival of plant species.
Objectives. The objectives are to characterize the flora of plant formations with Erythrina senegalensis and Erythrina sigmoidea in the Guiriko region and to analyze the current state of their populations in order to guide their conservation.
Method. Floristic and dendrometric data were collected following a targeted sampling after a survey of Erythrina plant formations. The plant formations selected for the study were the classified forests of Péni, Dindéresso, Kuinima, Kou, and the sacred woods of the sandstone hills of Kôrô.
Results. A weakly similar floristic diversity (Cs = 0.45) with 213 and 165 species, respectively, in the E. senegalensis formation and the E. sigmoidea formation was observed with Rubiaceae, Combretaceae, Fabaceae-ceasalpinioideae, Anacardiaceae, Fabaceae-faboideae, and Fabaceae-mimosoideae as dominant families. The most dominant biological types are the Phanerophytes. A regressive population dynamic characterized by a near absence of large-diameter individuals and adult individual densities of 25.48 individuals·ha-1 for E. senegalensis and 13.15 individuals·ha-1 for E. sigmoidea was observed. The low densities of juveniles (< 50 individuals·ha-1) and the low renewal potentials (< 2 individuals·ha-1) observed constitute a major risk to the survival of Erythrina populations. The populations of E. senegalensis are more subject to debarking (13.30% of plants), while that of E. sigmoidea is more subject to bush fires (88% of plants).
Conclusions. The renewal of these species is seriously compromised in the Guiriko region. Studies must be carried out on their regeneration modes, and special protection measures must be taken in conjunction with the populations to conserve and sustainably manage them.
Table of content
Received 28 March 2025, accepted 2 April 2026, available online 23 April 2026.
This article is distributed under the terms and conditions of the CC-BY License (https://creativecommons.org/licenses/by/4.0)
1. INTRODUCTION
1Africa harbours an exceptionally high level of biological diversity, which is essential for the future of the planet (Avikpo et al., 2017). A significant portion of this diversity is preserved within natural formations or protected areas. Plant species colonise these habitats and offer a variety of ecosystem services to humans and animals, contingent upon their capacity to adapt to environmental conditions and their ecological requirements. Several studies have shown that humans exploit a number of plant species for the various products and services they provide to local populations (Bélem et al., 2017; Kouadio et al., 2020). This situation has led to the overexploitation of multipurpose tree species with high socio-economic potential, thereby placing their populations in a regressive dynamic characterised by the scarcity or absence of juvenile individuals (Ouédraogo et al., 2006). Furthermore, several studies have reported that population dependence on natural formations affects the richness, structure, density, and basal area of woody species (Sahoo & Davidar, 2013; Sharma & Kant, 2014). Consequently, the phytodiversity of protected areas is under threat due to the combined effects of demographic and anthropogenic pressures, as well as climatic disturbances.
2In Burkina Faso, surveys conducted in protected areas have revealed a list of species that are overexploited due to their high value for local populations (Mbayngone & Thiombiano, 2011; Zerbo et al., 2022). These include Erythrina senegalensis DC. and Erythrina sigmoidea Hua, two species that are listed on the IUCN Red List. Field observations have revealed evidence of exploitation of Erythrina individuals. Furthermore, a scarcity or even absence of these two species has been observed in certain vegetation formations of the country despite the fact that they are endemic to these areas (Thiombiano et al., 2012; Arbonnier, 2019).
3According to the studies of Ouédraogo (2006), Ouoba et al. (2006), Traoré (2013) and Tindano (2016), E. senegalensis is widely distributed in the Sudanian zone of Burkina Faso. In contrast, E. sigmoidea is only referenced in the catalogue of vascular plants of Burkina Faso by Thiombiano et al. (2012), probably due to its restricted distribution or its status as an endangered species. The importance of these species, particularly in traditional medicine in West Africa, has been highlighted by several studies (Togola et al., 2008; Catarino et al., 2016; Frazao-Moreira, 2016; Dougnon et al., 2017; Sangaré et al., 2024). In Burkina Faso, E. senegalensis is used to treat pericardial pain (Ouoba et al., 2006), urinary tract infections (Kam et al., 2020), malaria, haemorrhoids, colds, coughs, yellow fever, mental health issues, and epilepsy (Sangaré et al., 2024). The species E. sigmoidea is used to treat malaria, haemorrhoids, yellow fever, eye ailments, and sexual weakness (Sangaré et al., 2024). However, such exploitation threatens the survival of these species, particularly E. sigmoidea, which appears to be declining in certain areas (Sangaré et al., 2024).
4Studies of the floristic and ecological characteristics of vegetation formations associated with a given species have often helped to restore declining populations and prevent their disappearance (Rabarison et al., 2013; Vitoekpon et al., 2018). Indeed, such studies provide essential information on the environmental conditions necessary for the survival, regeneration, and growth of species, as well as on interactions with other species within the ecosystem. They have also enabled the development of appropriate management plans that promote the restoration of degraded habitats and the implementation of effective conservation strategies. In the case of many threatened species, such as certain members of the Fabaceae family, understanding the vegetation formations with which they are associated has been essential for their reintroduction to ecosystems from which they were on the verge of disappearing (Mukenza et al., 2022). However, very limited information is available on the floristic and ecological characteristics of formations dominated by E. senegalensis and E. sigmoidea in Burkina Faso. For improved in situ and ex situ conservation of plant species, however, the adoption of sustainable harvesting techniques and a thorough understanding of their environment, dynamics, associations, and functioning are essential (Ouoba, 2006). Therefore, it is necessary to obtain scientific information on formations containing E. senegalensis and E. sigmoidea in Burkina Faso in order to identify favourable conditions for their preservation and prevent irreversible population decline. The present study was conducted within this context, with the overall objective of contributing to improved knowledge for the conservation and sustainable management of E. senegalensis and E. sigmoidea in Burkina Faso. Specifically, the study aims to characterise the floristic composition of formations containing E. senegalensis and E. sigmoidea in the Guiriko region and analyse the current status of their populations.
2. MATERIALS AND METHODS
2.1. Study environment
5The study was conducted in the classified forests of Dindéresso, Kuinima, Péni and Kou, and in the sacred woods of the sandstone hills of Kôrô in the Guiriko region (Figure 1). This area has a Sudano-Sahelian climate, characterised by a dry season from October to April and a rainy season from May to September. Mean annual rainfall ranges from 747.9 mm to 1,349.8 mm. The highest temperatures are mainly observed in March (37.38° C) and April (37.92 °C), while the lowest occur in January (18.04 °C) and September (21.54 °C) (ASECNA/Bobo-Dioulasso Meteorological Service, 2024). The soils are gravelly, sandy, sandy-loamy, and hydromorphic. Vegetation formations encountered include wooded savannahs, open forests, a few remnants of dense forests, and gallery forests along watercourses. Floristically, most of the woody species of the Sudanian domain are present, with the most frequently encountered being Afzelia africana Sm. ex Pers., Vitellaria paradoxa C.F.Gaertn., Detarium microcarpum Guill. & Perr., Parkia biglobosa (Jacq.) R.Br. ex G.Don, Lannea acida A.Rich., Combretum sp., Pterocarpus erinaceus Poir., Isoberlinia doka Craib & Stapf ex Holland, Anogeissus leiocarpa (DC.) Guill. & Perr., and Khaya senegalensis (Desr.) A.Juss. (Thiombiano & Kampmann, 2010).
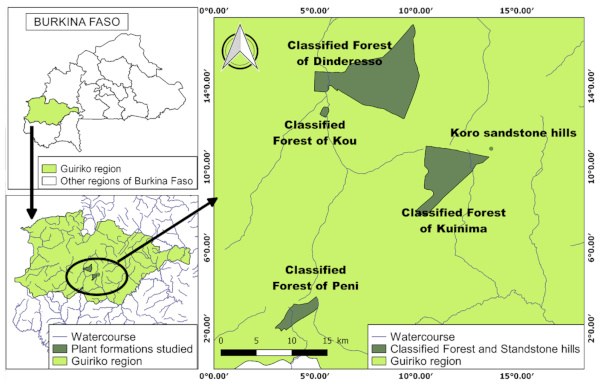
Figure 1. Location map of study sites – Carte de localisation des sites de l’étude.
Sources: BNDT, 2012 ; GPS surveys 2024.
2.2. Sampling
6The identification of Erythrina formations in Burkina Faso was made possible through a literature review of previous studies conducted in the country (Ouédraogo, 2008; Thiombiano et al., 2016; Tiendrébeogo, 2023), investigations with local communities and key informants, and field surveys. Formations were selected based on the presence of at least one studied Erythrina species, the relatively high density of individuals of that or those species, and the accessibility of the site.
7A targeted sampling approach involving the active search for individuals of Erythrina species was used to characterise their associated flora. This approach is suitable for the study of rare or threatened species (Acharya et al., 2000), although it does not allow for landscape-scale inferences. Erythrina sigmoidea is rare with a highly fragmented distribution, while Erythrina senegalensis is threatened and also has a fragmented distribution. Random or systematic sampling would risk failing to detect them altogether.
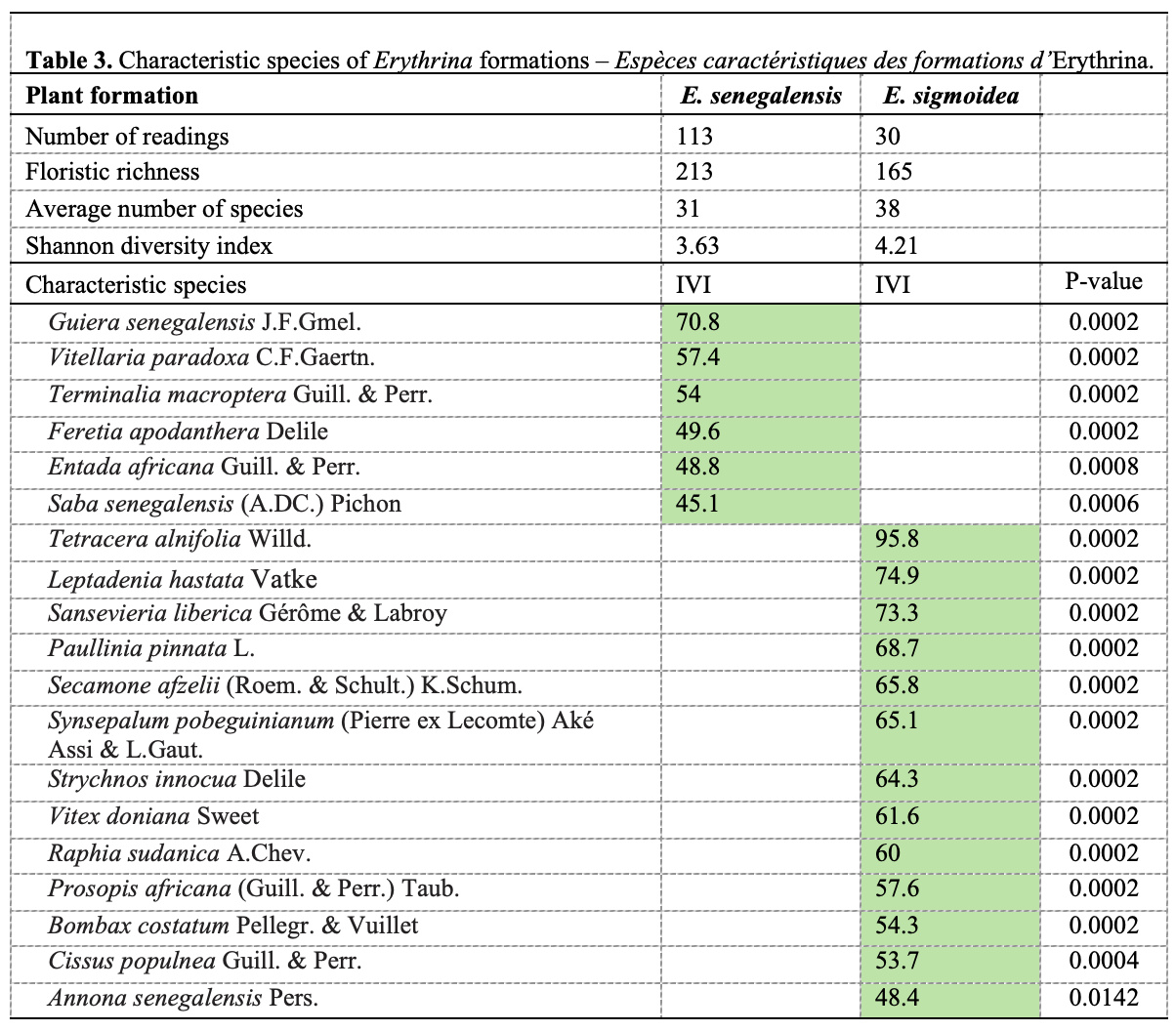
8Surveys were conducted by stratum and were targeted (Nacoulma, 2012) according to the presence of at least one individual of the genus Erythrina. For each Erythrina species, at least 30 sampling points were established around each Erythrina plant community, wherever possible. For the adult woody stratum, survey plots covered 900 m² (30 m × 30 m) in savannah formations and open forest formations, and 500 m² (50 m × 10 m) in gallery forests and riparian forest formations, following the recommendations of the Niamey workshop (Thiombiano et al., 2016). These plot sizes have been used in our study region by several authors (Ouoba, 2006; Tiendrébeogo et al., 2022). The same adult woody stratum plots were used to study the woody regeneration of Erythrina woody species (Kabré et al., 2020). This choice was made in consideration of the rarity of these species, for the regeneration study.
2.3. Data collection
9For each survey plot, a comprehensive list of species was compiled, and Erythrina individuals were recorded, counted, and measured for their diameter at breast height (DBH) at a height of 1.30 m above the ground and their total height (Ht). Individuals were classified as adults if their DBH was ≥ 5 cm and their Ht was ≥ 1.50 m, and as juveniles if their DBH was < 5 cm. To assess regeneration, juvenile individuals (DBH < 5 cm) were counted and grouped into height classes at 0.5 m intervals (Traoré, 2013). The health status of each Erythrina individual was recorded using the following code: 1) for apparently healthy individuals; 2) for pruned individuals; 3) for debarked individuals; 4) for uprooted individuals; and 5) for parasitised individuals.
2.4. Data analysis
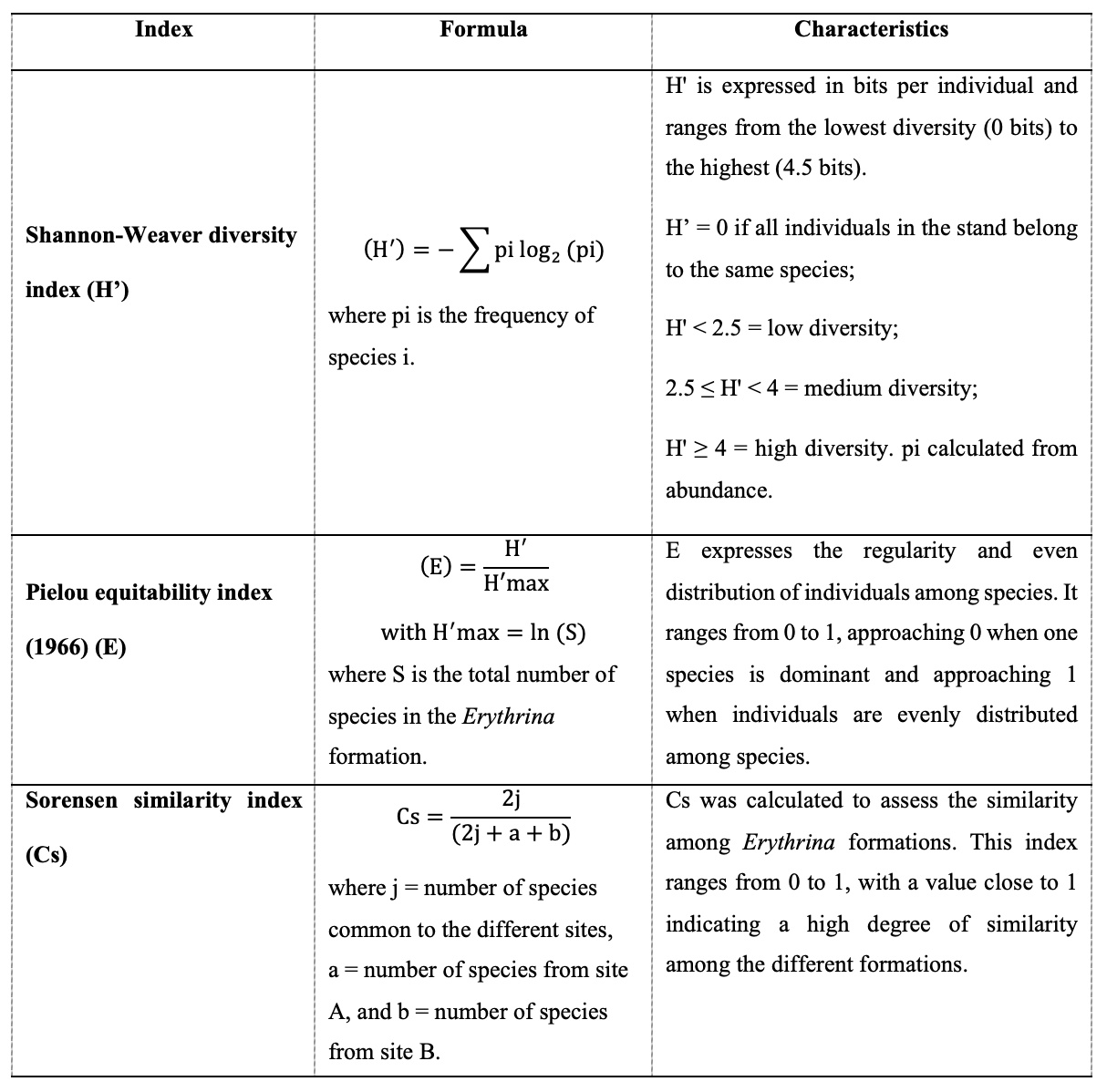
10Floristic richness and diversity. Species were identified, firstly, using the floras of Lebrun & Stork (1991–1997), Aubréville (1950), Hutchinson & Dalziel (1954–1972), and the botanical guide by Arbonnier (2019). Then, a list of the recorded species was compiled. These were subsequently classified by genus, family, biological type, and phytogeographical type. The spectra of families, biological types, and phytogeographical characteristics were presented in graphical form using Excel 2019. The same Excel spreadsheet was then used to calculate the diversity indices presented below:
11To identify the characteristic species of the ecosystems of the two plant species studied, an indicator species analysis was performed using the Indicator Value Index (IVI) method developed by Dufrêne & Legendre (1997). This analysis was conducted using PC-ORD software version 7.10 (Ouoba, 2006). The method combines species specificity and fidelity to provide an IVI index ranging from 0 to 100. The significance of the results was tested using Monte Carlo permutation tests (n = 4,999) at a significance level of 0.05.
12Dendrometric and structural characterisation of Erythrina populations. A dendrometric and structural assessment was performed by calculating the density (D) of individuals of each Erythrina species using the formula , where n is the total number of individuals of the Erythrina species considered, and S is the surface area of the plot (in m²).
13The diameter and height structures of the Erythrina populations were determined for each Erythrina species. These were expressed as the distribution of individuals across diameter classes starting from 5 cm (Traoré, 2013), and height classes starting from 2 m, fitted to a three-parameter Weibull distribution. This Weibull distribution was chosen due to the high variability of its shapes depending on the values taken by its parameters. Its probability density function, f, is given by the following formula :
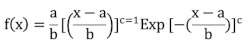
14with x = tree diameter ; f(x) = probability density value ; a = position parameter: it is equal to 0 if all trees categories are considered during the inventory, and it is non-zero if only trees with a diameter or height greater than or equal to a are considered. In this study, a is set to 5 cm for tree diameters and 1.50 m for the height of adult individuals ; b = scale or size parameter, related to the central value of the diameters of the trees in the population considered ; c = shape parameter linked to the diameter or height structure under consideration (Table 1). The Weibull parameters were calculated using Minitab 19 software and the structure graphs were established using Excel 2019.

15To detect the spatial patterns of E. senegalensis and E. sigmoidea individuals within stands, the Blackman index (IB) was calculated (Hamawa et al., 2018).
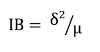
16with δ2 and μ, respectively the variance and mean density of the species in the plant formation considered. The distribution is regular (or uniform) if IB < 1; random if IB = 1 and aggregated (or clumped) if IB > 1.
17Erythrina sanitary status and population dynamics. Population status and dynamics were, firstly, assessed by counting healthy, debarked, pruned, uprooted, parasitized, and dead individuals of each species across the different vegetation formations. Next, the histograms of juvenile individuals were analysed according to height classes starting from 0.50 m (Ouédraogo, 2006; Traoré, 2013). Finally, the regeneration potential (d) was calculated as the ratio of juvenile to adult tree density. The following criteria, adapted from Gampiné & Boussim (1995), were applied : low regeneration if d ≤ 100 individuals·ha-1; medium regeneration if 100 < d ≤ 1,000; good regeneration if 1,000 < d ≤ 10,000; and very good regeneration if d > 10,000 individuals·ha-1.
3. RESULTS
3.1. Characterisation of the flora accompanying Erythrina species in the Guiriko region
18A total of 113 and 30 surveys were conducted for E. senegalensis and E. sigmoidea, respectively. Evaluation of the flora in Erythrina formations revealed a species richness ranging from 165 to 213, with high Shannon indices (3.63 bits and 4.21 bits) and Pielou’s evenness indices (0.79 and 0.87), indicating high diversity and regular distribution of individuals in the E. senegalensis and E. sigmoidea formations (Table 2). Sorensen's similarity index (Cs = 0.45) is well below 1, reflecting the low similarity between the different Erythrina formations.
19The floristic richness of the E. senegalensis formation is 213 species, including 116 woody species and 97 herbaceous species, distributed across 158 genera and 54 families. The most dominant families are Fabaceae-faboideae, followed by Rubiaceae, Malvaceae, Combretaceae, Fabaceae-ceasalpinioideae, Fabaceae-mimosoideae, Poaceae, and Lamiaceae. Families represented by no more than six species are grouped under “Other” (Figure 2).
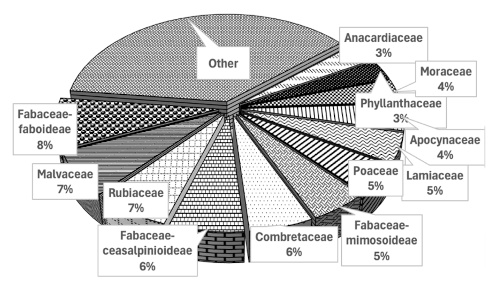
Figure 2. Spectrum of families in Erythrina senegalensis formations in the Guiriko region – Spectre des familles dans les formations d’Erythrina senegalensis dans la région de Guiriko.
20The flora of the E. sigmoidea formation comprises 165 species, including 114 woody species and 51 herbaceous species, distributed across 129 genera and 47 families. The most dominant families are Rubiaceae, Malvaceae, Fabaceae-faboideae, followed by Fabaceae-Ceasalpinioideae, Combretaceae, Poaceae, Fabaceae-Mimosoideae, and Lamiaceae. Families represented by no more than six species are grouped under “Other” (Figure 3).
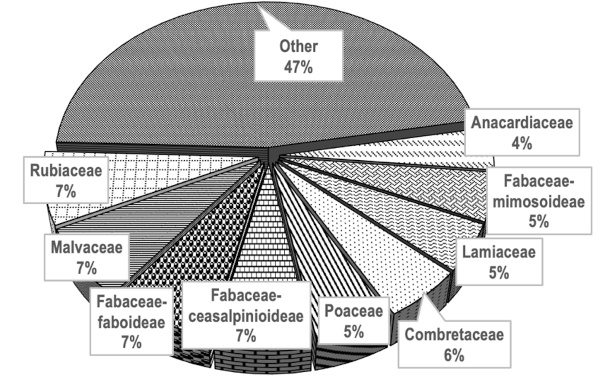
Figure 3. Spectrum of families in Erythrina sigmoidea formations in the Guiriko region – Spectre des familles dans les formations Erythrina sigmoidea dans la région de Guiriko.
21The distribution of biological types (Figure 4) shows that Erythrina formations are dominated by Phanerophytes, followed by Therophytes, Chamephytes, Geophytes and Hemicryptophytes.
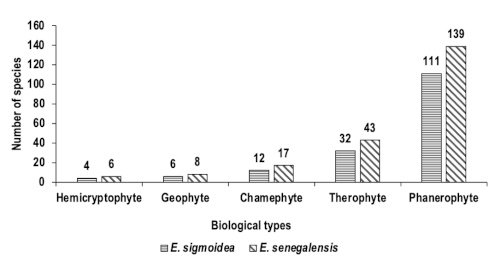
Figure 4. Biological spectrum of Erythrina senegalensis and E. sigmoidea formations in the Guiriko region – Spectre biologique des formations d’Erythrina senegalensis et d’E. sigmoidea dans la région de Guiriko.
22Phytogeographical types show a dominance of Afro-tropical (AT) species in Erythrina formations, followed by Sudano-Zambesian (SZ), Sudanian (S), Pantropical (Pan), transitional Guineo-Congolese and Sudano-Zambesian (GC-SZ), and Paleo-tropical (Pal) species. Guineo-Congolese, Sudano-Guinean, African-American (AM), Pluri-regional African (PA), and Pluri-regional (PT) species are in tiny proportions, with less than 10 species (Figure 5).
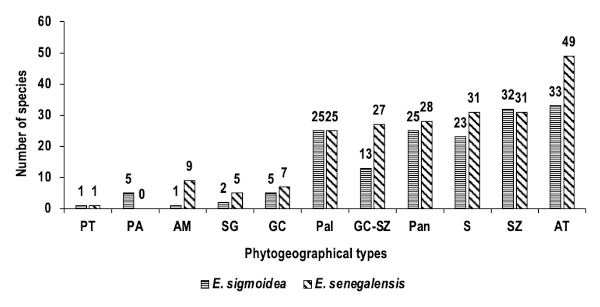
Figure 5. Phytogeographical spectrum of Erythrina senegalensis and E. sigmoidea formations in the Guiriko region – Spectre phytogéographique des formations d’Erythrina senegalensis et d’E. sigmoidea dans la région de Guiriko.
PT: Pluri-regional species – Espèces plurirégionales; PA: Pluri-regional African species – Espèces africaines plurirégionales; AM: African-American species – Espèces africano-américaines; SG: Sudano-Guinean species – Espèces soudano-guinéennes; GC: Guineo-Congolese species – Espèces guinéo-congolaises; Pal: Paleo-tropical species – Espèces paléo-tropicales; GC-SZ: Guineo-Congolese and Sudano-Zambesian species – Espèces guinéo-congolaises et soudano-zambéziennes; Pan: Pantropical species – Espèces pantropicales;S: Sudanian species – Espèces soudaniennes; SZ: Sudano-Zambesian species – Espèces soudano-zambéziennes; AT: Afro-tropical species – Espèces afro-tropicales.
3.2. Characteristic species of Erythrina formations
23The analysis revealed several species with strong ecosystem indicators for both species (Table 3). For E. senegalensis, species such as Guiera senegalensis J.F.Gmel. (IVI = 70.8; p = 0.0002), Vitellaria paradoxa C.F.Gaertn. (IVI = 57.4; p = 0.0002) and Entada africana Guill. & Perr. (IVI = 48.8; p = 0.0008) show a strong affinity. As for E. sigmoidea, Tetracera alnifolia DC. (IVI = 95.8; p = 0.0002), Leptadenia hastata Vatke (IVI = 74.9; p = 0.0002) and Sansevieria liberica Gérôme & Labroy (IVI = 73.3; p = 0.0002) stand out as major indicator species. However, these characteristic species do not necessarily indicate the presence of the Erythrina species studied.
3.3. Dendrometric characterisation of Erythrina populations
24The structural characteristics of Erythrina individuals (Table 4) show an average density of 25.48 ± 3.43 individuals·ha-1 in E. senegalensis formations, compared to 13.15 ± 1.95 individuals·ha-1 in the E. sigmoidea formation. The mean diameter at breast height (DBH) and mean total height were 12.42 ± 6.15 cm and 6.26 ± 1.96 m, respectively, in the E. senegalensis population and 19.60 ± 11.27 cm and 5.34 ± 1.85 m in the E. sigmoidea population.
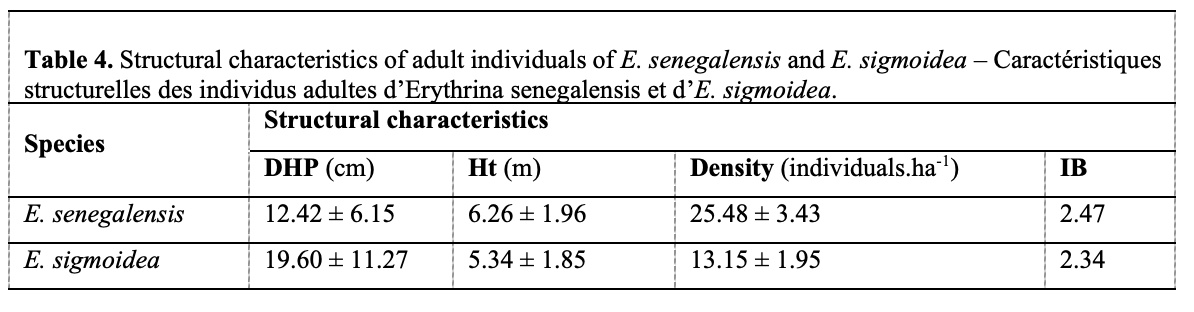
25Blackman index values indicate a clumped distribution of E. senegalensis and E. sigmoidea individuals (IB > 1) across the various formations.
26Diameter class distribution of Erythrina individuals. The diameter class distribution of E. senegalensis individuals (Figure 6a) shows an inverted J-shaped structure, which is characteristic of multispecific or uneven-aged stands (c < 1). As for the diameter class distribution of E. sigmoidea individuals (Figure 6b), it shows a positive or straight asymmetrical distribution (1 < c < 3.6), which is characteristic of monospecific stands with a predominance of young or small-diameter individuals.
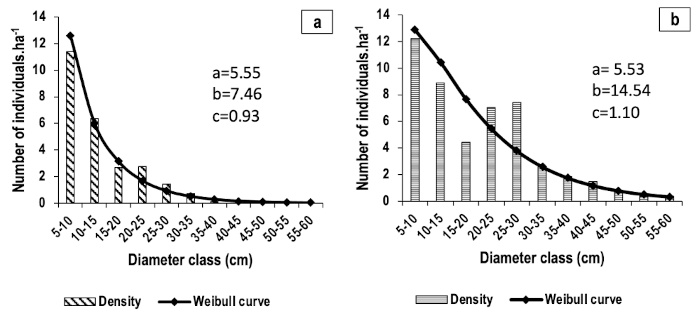
Figure 6. Diameter structures of adult individuals of E. senegalensis (a) and E. sigmoidea (b) – Structures diamétrales des individus adultes d’Erythrina senegalensis (a) et d’E. sigmoidea (b).
27Height-class distribution of Erythrina individuals. Analysis of the height-class distribution of Erythrina individuals in the identified formations (Figure 7) shows a positive or right asymmetric distribution structure (1 < c < 3.6), which is characteristic of populations with a predominance of young or low-height individuals.
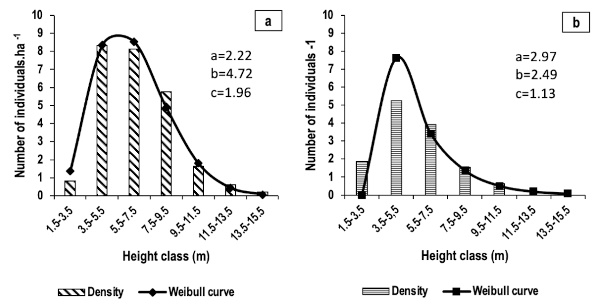
Figure 7. Height structures of adult individuals of E. senegalensis (a) and E. sigmoidea (b) – Structures de hauteur des individus adultes d’Erythrina senegalensis (a) et d’E. sigmoidea (b).
3.4. Erythrina population status and dynamics
28Health status of Erythrina populations. The health status of E. senegalensis individuals in the different formations shows that anthropogenic pressures are low, with a small proportion (≤ 20%) of individuals exhibiting signs of exploitation. These individuals are more susceptible to bark removal and parasitism (termite attack). In contrast, the population of E. sigmoidea is in poor health, as the majority of individuals (95%) are subjected to anthropogenic pressures with bushfires affecting 88% of them (Figure 8).
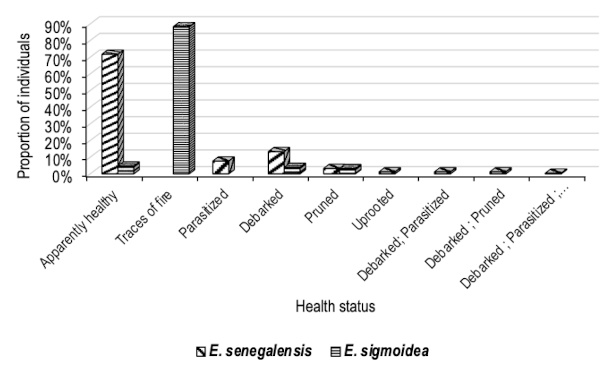
Figure 8. Health status of E. senegalensis and E. sigmoidea populations – État sanitaire des populations d’Erythrina senegalensis et d’E. sigmoidea.
29Juvenile Erythrina population dynamics. The distribution of juvenile Erythrina individuals by height class (Figure 9) shows “ inverted J-shaped ” structures, indicating an abundance of individuals in the ]0-0.5] m, ]0.51-1] m, and > 2 m classes.
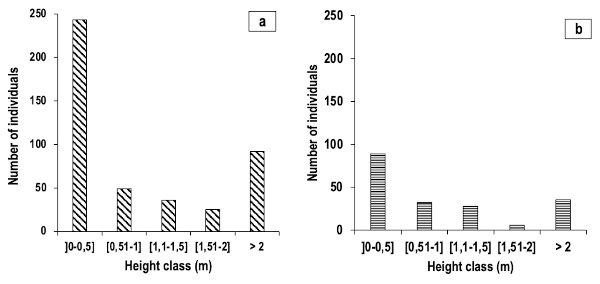
Figure 9. Juvenile population structures of E. senegalensis (a) and E. sigmoidea (b) – Structures des populations juvéniles d’Erythrina senegalensis (a) et d’E. sigmoidea (b).
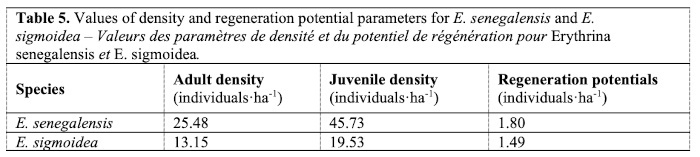
30Very low juvenile densities (Table 5) were observed for both species, with 25.48 individuals·ha-1 for the adult population of E. senegalensis and 45.73 individuals·ha-1 for its juvenile population. The adult density of E. sigmoidea is 13.15 individuals·ha-1, while the juvenile population density is 19.53 individuals·ha-1. The regeneration potentials are 1.80 individuals·ha-1 and 1.49 individuals·ha-1 for the E. senegalensis and E. sigmoidea populations, respectively.
4. DISCUSSION
31Dendrometric characterization of Erythrina populations in the Guiriko region revealed low populations densities (<< 100 individuals·ha-1) and average diameters of 12.42 ± 6.15 cm for E. senegalensis and 19.60 ± 11.27 cm for E. sigmoidea, with average heights of 6.26 ± 1.96 m and 5.34 ± 1.85 m, respectively. The diameter class distribution of E. senegalensis and E. sigmoidea individuals clearly reveals the predominance of young or small diameters individuals. The analysis of the distribution in height class shows an abundance in classes below 7 m compared to classes greater than or equal to 7 m. This form of distribution observed with the diameter classes of individuals of E. senegalensis and E. sigmoidea corroborates the findings reported by Hamawa et al. (2018) in Cameroon; Glèlè Kakaï & Sinsin (2009), Natta et al. (2011), Yehouenou Tessi et al. (2012), Dicko et al. (2017) in Benin, respectively, on the populations of Haematostaphis barteri, Isoberlinia spp., Pentadesma butyracea, Antiaris toxicaria and Lophira lanceolata. The low densities observed here are lower than those reported by Kabré et al. (2020) for Ziziphus mauritiana populations in eastern region of Burkina Faso. This could be explained by the strong anthropogenic pressure resulting from the removal of roots and bark and cutting. This finding corroborates the work of Nacoulma et al. (2011) and Kabré et al. (2020) who demonstrated that anthropogenic pressures reduce the density of Prosopis africana, Pterocarpus erinaceus and Ziziphus mauritiana in the eastern region of Burkina Faso. The low values of the average diameter and height of Erythrina individuals in the Guiriko region may reflect the shrubby nature of these species. Indeed, according to the classification of Arbonnier (2019), these species are generally shrubs reaching a height between 4 and 4.5 m, rarely exceeding 7 m. However, this structure cannot only be derived from the behavior of the species; it could also be linked to anthropogenic pressure. The development of young individuals is disrupted by their disappearance due to excessive exploitation of the species, difficult pedoclimatic conditions, grazing and bushfires. This would weaken the population. Nevertheless, in order to maintain itself, the population requires a significant proportion of young individuals to be incorporated into the adult population (Bationo et al., 2001; Hamawa et al., 2018). According to Ouédraogo et al. (2006), the predominance of young individuals is linked to disturbances or vulnerabilities at certain stages of its development. The absence of adult individuals would also affect the recovery of the population due to a lack of seeds (Mapongmetsem et al., 2011).
32Analysis of the health status of the populations shows that there are low proportions of healthy individuals among the adult population of E. sigmoidea, reflecting the vulnerability of the species in its natural habitats. This vulnerability is largely caused by bushfires (88% of individuals) in the sacred woods of the sandstone hills of Kôrô, the only site at which the species has been found. The low density observed at this site could be explained by grazing, fires and difficult soil conditions, which do not favour the proper development of individuals. In contrast, the low proportion of harvested individuals in the adult population of E. senegalensis indicates that the species is in good condition in its natural habitats. Erythrina senegalensis sites, such as the classified forests of Dindéresso, Kou, Kuinima, and Péni, as well as the sacred groves of the Kôrô sandstone hills, benefit from a degree of protection that mitigates the species' exposure to anthropogenic pressures.
33In the E. senegalensis and E. sigmoidea formations, regeneration dynamics reveal a high number of individuals in the first and last height classes, respectively. This could explain the good regeneration capacity of these species but the difficulty of survival, as the other intermediate classes contain few juvenile individuals. Renewal potential is also very low (<< 100 individuals·ha-1). This difficulty of survival could be explained by the types of habitats occupied by these species, as well as the challenging site and soil conditions found there. Habitats identified for E. senegalensis include gallery forests, riparian belts, hilltops, and bottoms on rocky soils and shrub savannas on clay-gravel soils. As for E. sigmoidea, gallery forests on rocky soils have been identified. These habitats, with their challenging environmental conditions, combined with bushfires and grazing, can compromise regeneration dynamics. Furthermore, as these habitats are periodically flooded, seeds can be washed away by runoff or rot due to excessive presence of water.
34The E. senegalensis formation distributed in the Guiriko region has a very high floristic richness, with 213 species in 158 genera and 54 families. It is only slightly similar (Cs = 0.45) to the E. sigmoidea formation, which comprises 165 species in 129 genera and 47 families. The low level of similarity and the richness of the E. senegalensis formation could be explained by the diversity of habitats occupied by the species such as shrubby and wooded savannahs, forest galleries and riparian cordons, compared to E. sigmoidea, which is only found in forest galleries and riparian cordons. Alternatively, this could be due to the large number of plant formations (5 plant formations) sheltering the species, compared to E. sigmoidea, which was only recorded in a single plant formation. Furthermore, the low richness of the E. sigmoidea formation can also be explained by the topographic homogeneity of the study area. According to Bridgewater et al. (2003) and Mbayngone (2008), the greatest floristic diversity is associated with environmental heterogeneity or habitat diversity. The dominance of the Fabaceae-Faboideae, Rubiaceae, Malvaceae, Combretaceae, Fabaceae-Ceasalpinioideae, Fabaceae-Mimosoideae and Poaceae families over other families is a characteristic of African savannahs (Ouoba, 2006; Nacoulma, 2012). Consequently, the vegetation of Erythrina formations has retained its original flora despite the observed anthropogenic and grazing pressures.
35The biological spectra of the E. sigmoidea and E. senegalensis formations are characterised by the dominance of phanerophytes (111 and 148 species, respectively), followed by therophytes (32 and 46 species, respectively). This reflects the fact that our study area belongs to the Sudanian zone of Burkina Faso (Ouoba, 2006; Thiombiano & Kampmann, 2010). The strong presence of phanerophytes offers hope for the natural restoration of degraded plant formations (Issifou et al., 2019). According to Havyarimana et al. (2013), phanerophytes dominate less disturbed environments, whereas chamephytes colonise stressful environments. This demonstrates that the protection status assigned to these studied ecosystems mitigates the effects of human impact. The "classified" status of Dindéresso, Kou, Kuinima and Péni forests and the presence of places of worship within the formation of the sandstone hills of Kôrô, could help to reduce anthropic pressures in these areas because cutting and other exploitation of species are prohibited there. The high proportion of phanerophytes in these plant formations could facilitate the survival of Erythrina species by creating a humid and relatively mild microclimate with only a slightly pronounced unfavorable season.
36Analysis of the phytogeographic spectra of the E. sigmoidea and E. senegalensis formations reveals an abundance of African pluriregional species (Afrotropical, Sudanian, Sudano-Zambézian, Guineo-Congolese, species common to the last two taxa) followed by species with a wide distribution (Pantropical, Paleotropical). The high proportion of Guineo-Congolese, Sudano-Zambézian species and species common to the last two taxa confirms that the study environment belongs to the Sudanian zone. The abundance of species with wide (Pantropical and Paleotropical) and continental (Afrotropical) distributions after the Sudanian species indicates that the E. senegalensis and E. sigmoidea formations belong to the disturbed Sudanian domain (Sinsin, 2001; Melom et al., 2015).
37The indicator species analysis approach made it possible to accurately distinguish the species most representative of each Erythrina formation. The results obtained demonstrate significant floristic differentiation between the two formation types, corroborating the previously observed weak similarity as determined by Sorensen's index (Cs = 0.45). Several species in the E. senegalensis formation have high and significant IVI values, reflecting their strong specificity and fidelity to this environment. These include Guiera senegalensis, Vitellaria paradoxa, Terminalia macroptera and Entada africana, which are known for their association with Sudanian savannahs (Thiombiano et al., 2012). These characteristic species confirm the ecological profile of the E. senegalensis formation, which is dominated by heliophilous species that can withstand anthropogenic disturbance. In the E. sigmoidea formation, the indicator species are even more specific, with very high IVI values: Tetracera alnifolia (95.8), Leptadenia hastata (74.9), Sansevieria liberica (73.3), and Strychnos innocua (64.3). These species are associated with more closed or humid formations (forest galleries) reflecting an ecological environment that is distinct from that of E. senegalensis. This corroborates field observations of the more restricted and specialised ecological distribution of E. sigmoidea, which is limited to more fragile and localised habitats. The abundance of multi-regional African species and/or those with a wide distribution indicates broad ecological tolerance to variations in climate and soil, and sometimes to non-extreme disturbances. These abundances could contribute to reducing the vulnerability of Erythrina species.
5. CONCLUSIONS
38This study of the Erythrina species present in the Guiriko region reveals that their flora is rich and diverse, with an abundance of phanerophytes over therophytes. Phytogeographical spectra reveal an abundance of Pantropical, Paleotropical, Afrotropical, and Sudanian species, indicating that the E. senegalensis and E. sigmoidea formations belong to the disturbed Sudanian domain. Furthermore, a study of their availability reveals the presence of specimens of E. sigmoidea in the Guiriko region, specifically in the Houet province, on the sandstone hills of Kôrô. However, the structural characteristics of Erythrina individuals indicate that there are young populations subject to several anthropogenic pressures, with a significant proportion of apparently healthy E. senegalensis individuals and a more fire-prone E. sigmoidea population. Therefore, the different plant formations, by their status, contribute to the preservation of the species. In order not to compromise the conservation of Erythrina species, it is recommended that site protection against fires, logging and the uncontrolled exploitation of sensitive areas is strengthened, and that awareness of E. senegalensis debarking is raised, as well as the enrichment of identified favourable habitats through planting. For domestication and conservation purposes, particular attention must be given to investigating the modes of reproduction of E. senegalensis and E. sigmoidea.
Bibliographie
Acharya B., Bhattarai G., De-Gier A. & Stein A., 2000. Échantillonnage systématique adaptatif par grappes pour l'évaluation des espèces d'arbres rares au Népal. Écologie Gestion For., 137(1-3), 65-73.
Arbonnier M., 2019. Arbres, arbustes et lianes des zones sèches d’Afrique de l’ouest. 4è ed. Versailles, France : Éditions Quæ.
Aubréville A., 1950. Flore forestière soudano-guinéenne. Paris : Société d'Éditions Géographiques, Maritimes et Coloniales.
Avikpo D.J. et al., 2017. Impact des caractéristiques de la végétation sur la diversité d’usages des plantes autour de deux grandes forêts classées et d’une réserve botanique au Sud-Bénin. Eur. Sci. J., 13(30), 376-394, doi.org/10.19044/esj.2017.v13n30p376
Bationo B.A., Ouédraogo S.J. & Guinko S., 2001. Longévité des graines et contraintes à la survie des plantules d'Afzelia africana Sm. dans une savane boisée du Burkina Faso. Ann. For. Sci., 58(1), 65-75.
Bélem M., Nabaloum M. & Yaméogo J., 2017. Strategy of conservation and protection of wild edible plants diversity in Burkina Faso. Anadolu J. Aegean Agric. Res. Inst., 27(2), 82-90.
Bridgewater S. et al., 2003. A preliminary floristic and phytogeographic analysis of the woody flora of seasonally dry forest in northern Peru. Candollea, 58, 129-148.
Catarino L., Havik P.J. & Romeiras M.M., 2016. Medicinal plants of Guinea-Bissau: therapeutic applications, ethnic diversity and knowledge transfer. J. Ethnopharmacol., 183, 71-94, doi.org/10.1016/j.jep.2016.02.032
Dicko A. et al., 2017. Typology and structural characterization of Lophira lanceolata populations in Benin (West Africa). Rev. Cames, 5(1), 36-43.
Dufrêne M. & Legendre P., 1997. Species assemblages and indicator species : the need for a flexible asymmetrical approch. Ecol. Monogr., 63(3), 345-366, doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
Dougnon T.V. et al., 2017. Traditional treatment of human and animal salmonelloses in Southern Benin: knowledge of farmers and traditherapists. Vet. World, 10(6), 580-592, doi.org/10.14202/vetworld.2017.580-592
Frazao-Moreira A., 2016. The symbolic efficacy of medicinal plants: practices, knowledge and religious beliefs amongst the Nalu healers of Guinea-Bissau. J. Ethnobiol. Ethnomed., 12(24), 1-15, doi.org/10.1186/s13002-016-0095-x
Glèlè Kakaï R. & Sinsin B., 2009. Structural description of two Isoberlinia dominated vegetation types in the Wari-Maro Forest Reserve (Benin). S. Afr. J. Bot., 75, 43-51, doi.org/10.1016/j.sajb.2008.07.003
Hamawa Y., Balna J. & Souaré K., 2018. Structure écologique et production fruitière de Haematostaphis barteri Hook. F en la zone sahélienne du Cameroun. J. Appl. Biosci., 130, 13232-13243, doi.org/10.4314/jab.v130i1.10
Havyarimana F. et al., 2013. Diversité et distribution d’abondances des plantes d’un écosystème protégé dans un paysage anthropisé : cas de la Réserve Naturelle Forestière de Bururi, Burundi. Tropicultura, 31(1), 28-35.
Hutchinson J. & Dalziel J.M., 1954, 1958, 1963, 1968, 1972. Flora of West Tropical Africa (Vol. 1 à 3 : Vol. 1, tomes 1 et 2, Vol. 2, Vol. 3, tomes 1 et 2). London : Crown Agents for Overseas Governments and Administrations.
Issifou M.Y., Toko I.I. & Djalilou M.A., 2019. Caractérisation de la dégradation à travers la diversité floristique et la structure de la végétation dans le Bassin Moyen de la Sota au Nord-Bénin. Int. J. Progressive Sci. Technol., 18(1), 53-70.
Kabré B., Belem/Ouédraogo M. & Ouédraogo A., 2020. Variabilités structurale et floristique des populations du jujubier, Ziziphus mauritiana Lam. en zone semi-aride du Burkina Faso (Afrique de l’Ouest). Sci. Nat. Appl., 1(29), 135-149.
Kam S.E. et al., 2020. Ethnobotanical survey of plants used by traditional healers for treatment of urinary infections in Hauts-Bassins areas of Burkina Faso. Int. J. Sci. Res., 9(5), 1113-1118, doi.org/10.21275/SR20506024028
Kouadio Y. J-C. et al., 2020. Disponibilité des plantes utilitaires dans la zone de conservation de biodiversité du barrage hydroélectrique de Soubré, Sud-Ouest, Côte d’Ivoire. Afr. Sci., 16(6), 65-74.
Lebrun J.-P. & Stork L.A. (1991, 1992, 1995, 1997). Énumération des plantes à fleurs d’Afrique tropicale (Vol. 1 ; Vol. 2 ; Vol. 3 ; Vol. 4). Genève, Suisse : Conservatoire et Jardin botaniques de la Ville de Genève.
Mapongmetsem P.M. et al., 2011. Impact des systèmes d'utilisation des terres sur la conservation de Vitellaria paradoxa Gaerten. F. (Sapotaceae) dans la région des savanes soudano-guinéennes. Int. J. Environ. Stud., 68(6), 851-872, doi.org/10.1080/00207233.2011.587259
Mbayngone E., 2008. Flore et végétation de la réserve partielle de faune de Pama, sud-est du Burkina Faso. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Mbayngone E. & Thiombiano A., 2011. Dégradation des aires protégées par l’exploitation des ressources végétales : cas de la réserve partielle de faune de Pama, Burkina Faso (Afrique de l’Ouest). Fruits, 66(3), 187-202, doi.org/10.1051/fruits/2011027
Melom S. et al., 2015. Caractéristiques floristique et écologique des formations végétales de Massenya au Tchad (Afrique centrale). J. Anim. Plant Sci., 25(1), 3799-3813.
Mukenza M.M. et al., 2022. L’exploitation incontrôlée de Pterocarpus tinctorius Welw., une menace sur les écosystèmes forestiers et la sécurité alimentaire dans le territoire de Kasenga en République Démocratique du Congo (R.D.C.). In : Schmitz S. & Nicola S., eds. Actes du Colloque international « Changements globaux et gestion de la transition : au singulier ou au pluriel ? », 20-21 octobre 2022, Université de Liège, Belgique. Liège, Belgique : Atelier des Presses, 89-90.
Nacoulma B.M.I., 2012. Dynamique et stratégies de conservation de la végétation et de la phytodiversité du complexe écologique du parc national du W du Burkina Faso. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Nacoulma B.M.I., Traoré S., Hahn K. & Thiombiano A., 2011. Impact of land use types on population structure and extent of bark and foliage harvest of Afzelia africana and Pterocarpus erinaceus in Eastern Burkina Faso. Int. J. Biodivers. Conserv., 3(3), 62-72.
Natta A.K. et al., 2011. Typologie et structure des populations naturelles de Pentadesma butyracea dans la zone soudano-guinéenne du Bénin. Ann. Sci. Agron., 15, 137-152.
Ouédraogo A., 2006. Diversité et dynamique de la végétation ligneuse de la partie orientale du Burkina Faso. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Ouédraogo A., Thiombiano A., Hahn-Hadjali K. & Guinko S., 2006. Structure du peuplement juvénile et potentialités de régénération des ligneux dans l’Est du Burkina Faso. Étude Flore Vég. Burkina Faso, 10, 17-24.
Ouédraogo M., 2008. Les galeries forestières de la Réserve de la Biosphère de la Mare aux Hippopotames du Burkina Faso : caractéristiques, dynamique et ethnobotanique. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Ouoba P., 2006. Flore et végétation de la forêt classée de Niangoloko, Sud-ouest du Burkina Faso. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Ouoba P., Lykke A.M., Boussim I.J. & Guinko S., 2006. La flore médicinale de la Forêt classée de Niangoloko (Burkina Faso). Étude Flore Vég. Burkina Faso, 10, 5-16.
Rabarison H. et al., 2013. Évaluation du risque d’extinction des Cedrelopsis (Rutaceae) de Madagascar. Scripta Bot. Belg., 50, 41-49.
Sahoo S. & Davidar P., 2013. Effect of harvesting pressure on plant diversity and vegetation structure of Sal forests of Similipal Tiger Reserve, Odisha. Trop. Ecol., 54(1), 97-107.
Sangaré I., Tiendrébeogo N.R. & Ouoba P., 2024. Usages, perceptions des facteurs de dégradations et stratégies locales de conservation d’Erythrina senegalensis A.DC. et d’Erythrina sigmoidea Hua dans la région des Hauts-Bassins (Burkina Faso). Int. J. Biol. Chem. Sci., 18(5), 1815-1831, doi.org/10.4314/ijbcs.v18i5.16
Sharma N. & Kant S., 2014. Vegetation structure, floristic composition and species diversity of woody plant communities in sub-tropical Kandi Siwaliks of Jammu, J & K, India. Int. J. Basic Appl. Sci., 3(4), 382-391, doi:10.14419/ijbas.v3i4.3323
Sinsin B., 2001. Formes de vie et diversité spécifique des associations de forêt claires du nord du Benin. Syst. Geogr. Plants, 71, 873-888, doi.org/10.2307/3668725
Thiombiano A. & Kampmann D., 2010. Atlas de la biodiversité de l’Afrique de l’Ouest. Tome II. Burkina Faso. Ouagadougou; Frankfurt/Main, Allemagne : Projet BIOTA Afrique.
Thiombiano A. et al., 2012. Catalogue des plantes vasculaires du Burkina Faso. Boissiera, 65.
Thiombiano A. et al., 2016. Méthodes et dispositifs d’inventaires forestiers en Afrique de l’Ouest : état des lieux et propositions pour une harmonisation. Ann. Sci. Agron., 20, 15-31.
Tiendrébeogo N.R., 2023. Phytodiversité, végétation et services écosystémiques de la Forêt classée de Péni au Sud-Ouest du Burkina Faso. Thèse de doctorat : Université Nazi BONI (Burkina Faso).
Tiendrébeogo N.R. et al., 2022. Phytodiversity and vulnerability of protected areas in Burkina Faso : case of Péni classified forest. J. Geosci. Env. Prot., 10, 204-223, doi.org/10.4236/gep.2022.1012012
Tindano E., 2016. État et dynamique de la végétation et de la flore des inselbergs du Burkina Faso. Thèse de doctorat : Université Ouaga I Pr Joseph Ki-Zerbo (Burkina Faso).
Togola A. et al., 2008. Ethnopharmacological uses of Erythrina senegalensis: a comparison of three areas in Mali, and a link between traditional knowledge and modern biological science. J. Ethnobiol. Ethnomed., 4(6), doi.org/10.1186/1746-4269-4-6
Traoré L., 2013. Influence du climat et de la protection sur la végétation ligneuse de la partie occidentale du Burkina Faso. Thèse de doctorat : Université de Ouagadougou (Burkina Faso).
Vitoekpon I., Adomou A.C. & Oumorou M., 2018. Statut de conservation des peuplements à Mansonia altissima dans la forêt sacrée d’Ewé-Adakplame au Bénin. Rev. Ivoirienne Sci. Technol., 32, 350-370.
Yehouenou Tessi R.D., Akouehou G.S. & Ganglo J.C., 2012. Caractéristiques structurales et écologiques des populations de Antiaris toxicaria (Pers.) Lesch et de Ceiba pentandra (L.) Gaertn dans les forêts reliques du Sud Bénin. Int. J. Biol. Chem. Sci., 6(6), 5056-5067.
Zerbo I. et al., 2022. Ethnobotanical knowledge and conservation of Bombax costatum Pellegr. & Vuillet: an over-exploited savanna tree species. Trees For. People, 10, 100356, doi.org/10.1016/j.tfp.2022.100356






