- Accueil
- volume 9 (2005)
- numéro 3
- Variability of the microbial abundance of a kefir grain starter cultivated in partially controlled conditions
Visualisation(s): 3080 (15 ULiège)
Téléchargement(s): 131 (1 ULiège)
Variability of the microbial abundance of a kefir grain starter cultivated in partially controlled conditions

Notes de la rédaction
Received 7 September 2004, accepted 11 May 2005
Résumé
Variabilité de l’abondance microbienne d’un ferment de kéfir traditionnel cultivé en conditions partiellement contrôlées. La variabilité de l’abondance en lactobacilles, en coques lactiques et en levures de grains de kéfir cultivés dans des conditions partiellement contrôlées – manipulations de renouvellement du lait à température ambiante et incubation en étuve contrôlée - a été quantifiée. Exprimée par l’écart type relatif géométrique d’énumérations répétées dans le temps, elle était respectivement de 28 %, 443 % et 35 % pour chacun des trois groupes microbiens. L’origine de la variabilité microbienne observée a été attribuée au développement d’un milieu hétérogène autour des grains pendant la fermentation et, pour les coques lactiques, plus encore aux conditions initiales de fermentation, probablement des paramètres liés au renouvellement du lait (température ambiante de la pièce, temps d’attente avant incubation).
Abstract
The variability of the abundance in lactobacilli, lactic acid streptococci and yeasts of a kefir grain starter cultivated in partially controlled conditions – milk renewal at room temperature and incubation at defined temperature - was quantified. Expressed by the geometric relative standard deviations of counts repeated over time, it was respectively of 28%, 443% and 35% for each of the three microbial groups. The origin of the microbial variability observed was ascribed to the heterogeneous medium developed around the grains during fermentation and, for the lactic acid streptococci, even more to the initial conditions of fermentation, probably to parameters linked to milk renewal (room temperature, waiting time at room temperature before incubation).
Table des matières
1. Introduction
1Kefir grains are those Caucasian originated dairy starters that look like cauliflower florets. These grains consist of a complex set of lactic acid bacteria, yeasts and, sometimes, acetic bacteria embedded in a specific polysaccharide matrix. Kefir grains have the property to perpetuate, that is growing and multiply while maintaining a stable population. This constancy of the grain microbial composition is a generally accepted feature (Garrote et al., 1998; Kuo, Lin, 1999; Simova et al., 2002) that stay vague.
2The aim of this work is to assess the standard deviation around it for the main microbial groups of a kefir grain starter cultivated in partially controlled conditions and to point out the sources of variation.
2. Materials and methods
2.1. Origin and maintenance of the kefir grain starter
3The kefir grain starter comes from the collection of the Walloon Agricultural Research Centre, Department Quality of agricultural products (Gembloux, Belgium). It has been collected in the 1990’s from a Belgian household. Since then, it has been propagated in sterilised cow milk renewed daily during the working days. At every transfer in fresh tempered milk, the grains are rinsed with sterile water. Handlings of milk renewal occur at room temperature and grains are then incubated without shaking at 22°C. Once a week, the grain: milk ratio is adjusted to 20% (w/v).
4For the experiment, two sub-samples of the kefir grain starter were cultivated in this way in standardised containers. At the time of grain: milk ratio adjustment, the weight of the rinsed and dripped kefir grains was reduced to 57 g ± 3 g. Care was taken to limit the weight difference between duplicates to less than 2 g.
2.2. Microbial composition of the kefir grain starter
5Lactobacilli, lactic acid streptococci and yeasts in the two cultures were counted weekly for seven weeks. Grains were harvested for counting at the time of grain: milk ratio adjustments.
2.3. Microbial counts
6A first 1:9 (w/v) dilution was prepared by dispersing 12 g ± 3 g of rinsed and dripped grains in the appropriate volume of Ringer solution with an Ultra-turrax T25 (IKA-Labortechnik, Germany). Subsequent serial dilutions were plated on MRS-Agar, on M 17-Agar and on YGC-Agar for enumeration of respectively the lactobacilli, the lactic acid streptococci and the yeasts (all the media were purchased from Merck). Plates were incubated anaerobically during three days at 37°C for the lactobacilli, aerobically during respectively two days at 37°C and five days at 22°C for the lactic acid streptococci and the yeasts. The exclusive presence of yeasts on the yeast-mould specific medium was examined through colony morphology.
2.4. Accuracy of the microbial counting methods
7Accuracy of the microbial counts was determined on seven replicates coming from a single batch of grains.
2.5. Statistical analysis
8Standard deviations between duplicates (repeatability of measures between duplicates at the same time) and between weeks (reproducibility of measures over time) were deduced from the variance components calculated with the VARCOMP procedure of the SAS software (SAS Institute, 1994).
2.6. Visualisation of microbial development with a chromogenic substrate
9Two grains of about 4 g each were cultivated in parallel during 24 hours in milk added with 5-bromo-4-chloro-3-indolyl-b-D-galactopyranoside (X-gal) at 0.4 mg ml-1. One of the cultures was kept unshaken during the growing while the other was shaken at 140 rotations per minute. They were both incubated at 22°C with a grain: milk ratio of 20% (w/v).
3. Results and discussion
10The geometric means of the microbial counts, expressed in CFU* g-1 of grains, were of 1.4 ¥ 108, 3.9 ¥ 104 and 1.1 ¥ 107 respectively for the lactobacilli, the lactic acid streptococci and the yeasts. These figures of microbial abundance in kefir grains are comparable to those published by Pintado et al. (1996).
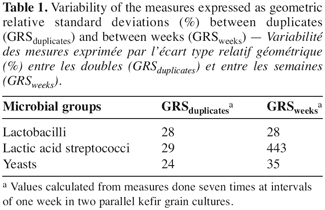
11The geometric relative standard deviations (GRS) of the counts, assigned to each of the two sources of variation: the duplicate cultures (GRSduplicates) and the successive weeks (GRSweeks), are shown for each microbial group in table 1. Due to the accuracy of the counting methods, the deviation between the two cultures and, even more, between weeks was undoubtedly biologically significant. The counting methods gave indeed measurement errors that, expressed as GRS, were as low as 9%, 7% and 4% for lactobacilli, lactic acid streptococci and yeasts respectively.
12To our knowledge, the literature on the microbial composition of kefir grains refers only to occasional measurements, not repeated over time, and only one study has been conducted with individual cultures of the same kefir grain starter (Simova et al., 2002). Calculated from the author’s data, the GRS for the cultures are 32%, 18% and 57% respectively for lactobacilli, lactic acid streptococci and yeasts. These figures express a variation comparable to that observed in our study, but its biological significance is uncertain.
13Table 1 reveals that the growth of the three microbial groups varied between the cultures. The growth of the lactic acid streptococci and, to a lesser extent, the yeasts showed even more unstable over time as the week-to-week deviation exceeds that between duplicates.
14In order to point out the origin(s) of the variations observed in microbial growth, the uncontrolled parameters were picked out and analysed.
15Among them, appeared the manual rinsing of the grains with a degree of rinsing being left to the operator’s discretion. The subjectivity associated with grain rinsing was however not a major source of microbial variation, as omitting this step in a parallel trial involving two other cultures did not in fact affect the grain microbial composition (data not show).
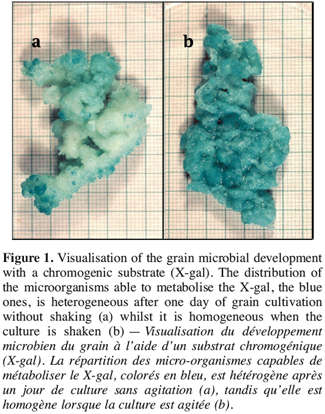
16Other possible sources of variation were the rising of grains to the surface and the syneresis of the milk that occurred during fermentation. These events generated a heterogeneous medium around the grains that was clearly variable: on removal from the incubator, some grains were surrounded by air while others by whey or curd and this varied from container to container. The relation between this observation and microbial variation implies that the heterogeneous medium generated during fermentation may influence the grain microbial growth. This latter hypothesis was confirmed with the aid of a chromogenic substrate revealing a galactosidase activity. As lactose is the main available carbohydrate source, the activity of this enzyme is indeed an indicator of the microbial growth for the lactose-metabolising organisms, let say the majority of the kefir grain microflora. Grain coming from the heterogeneous medium showed an uneven distribution of microorganisms with a galactosidase activity (Figure 1a), while it was evenly distributed throughout the all grain when it was incubated in a medium that was homogenised by shaking (Figure 1b). That this discrepancy in growing behaviour is due to an external origin was confirmed by giving evidence that the microorganisms from the uncoloured parts of the grain may have a galactosidase activity. Spreading smear preparations from these areas on a culture medium in fact produced colonies that all turned blue when drops of chromogenic substrate were added (data not show).
17Some last parameters that determined the initial fermentation conditions i.e. just before incubation could have been a source of variation. Indeed, the milk was temperate and renewed at ambient temperature, let say not regulated. Furthermore, due to the design of the experiment, cultures had to be allowed to stand for about three-quarters of an hour before being put into the incubator once the milk had been renewed. Although this duration is rather short if compared to the length of the one day incubation, it might have a significant influence on the evolution of the kefir grain microorganisms. This point was indirectly confirmed by figures obtained for the milk pH measured just before placing the cultures in the incubator. From one day to another, they varied in a range between 5.3 and 6.0, showing that the microbial activity was already important and irregular. The microorganisms responsible for primary acidification of the milk in kefir making are lactic acid streptococci (Koroleva, 1988), precisely those that showed the greatest variability of counts over time. Moreover, among them, lactococci are reputed to be heat-sensitive (Dellagio et al., 1994).
4. Conclusions
18Significant variation in quantitative microbial composition of a kefir grain starter occurred in partially controlled cultivation conditions. Variation in microbial abundance was observed for the three main microbial groups of the kefir grain starter, but, among them, the lactic acid streptococci showed by far the greatest variability.
19Events that come with kefir fermentation -grain rising and milk syneresis- were pointed out for being at the source of a microbial variability that affected almost equally the three microbial groups.
20The lactic acid streptococci were however more affected by another source of variability that occurred at the early beginning of the fermentation. This source was probably related to the room temperature and the waiting time of the containers before placing into the incubator. Those latter parameters were indeed not regulated.
21Acknowledgements
22We are grateful to Mohamed Echahraoui for his technical assistance.
Bibliographie
Dellagio F., de Roissart H., Torriani S., Curk MC., Janssens D. (1994). Taxonomie, métabolisme, croissance et génétique des bactéries lactiques. In de Roissart H., Luquet FM. (eds.). Bactéries lactiques. Uriage, France : Lorica vol. 1, p. 23–116.
Garrote GL., Abraham AG., De Antoni GL. (1998). Characteristics of kefir prepared with different grain: milk ratios. J. Dairy Res. 65, p. 149–154.
Koroleva NS. (1988). Kefir and kumys starters. Bull. Int. Dairy Fed. 227, p. 35–40.
Kuo CY., Lin CW. (1999). Taiwanese kefir grains: their growth, microbial and chemical composition of fermented milk. Aust. J. Dairy Technol. 54, p. 19–23.
Pintado ME., Da Sylva LJA., Fernandes PB., Malcata X., Hogg JA. (1996). Microbiological and rheological studies on Portuguese kefir grains. Int. J. Food Sci. Technol. 31, p. 15–26.
SAS Institute Inc. (1994). SAS/STAT User’s Guide. Cary, NC, USA: Statistical Analysis System Institute.
Simova E., Beshkova D., Angelov A., Hristozova Ts., Frengova G., Spasov Z. (2002). Lactic acid bacteria and yeasts in kefir grains and kefir made from them. J. Ind. Microbiol. Biotechnol. 28, p. 1–6.






